Articles
- Page Path
- HOME > Osong Public Health Res Perspect > Volume 7(6); 2016 > Article
-
Original Article
No Detection of Episomal or Integrated High-Risk Human Papillomavirus in Nonsmall Cell Lung Carcinomas among Korean Population - Jeong Eun Leea, Yong-Moon Leeb, In-Ock Seongb, Min-Woong Kangc, Choong Sik Leeb, Kyung-Hee Kimb
-
Osong Public Health and Research Perspectives 2016;7(6):356-359.
DOI: https://doi.org/10.1016/j.phrp.2016.10.004
Published online: October 21, 2016
aDivision of Pulmonology, Department of Internal Medicine, Chungnam National University, Daejeon, Korea
bDepartment of Pathology, Chungnam National University School of Medicine, Daejeon, Korea
cDepartment of Thoracic and Cardiovascular Surgery, School of Medicine, Chungnam National University, Daejeon, Korea
- ∗Corresponding author. phone330@cnu.ac.kr
• Received: July 30, 2016 • Revised: October 5, 2016 • Accepted: October 14, 2016
Copyright © 2016 Korea Centers for Disease Control and Prevention. Published by Elsevier Korea LLC.
This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
Abstract
-
Objectives
- High-risk human papillomavirus (hrHPV) is known to be a representative cancer-causing agent in the genital and head and neck regions. Many studies have detected hrHPV DNA in nonsmall cell lung carcinoma. However, hrHPV–etiologic correlation in nonsmall cell lung carcinoma remains unclear. This study is designed to determine the prevalence of episomal or integrated hrHPV DNA in nonsmall cell lung carcinoma among the Korean population.
-
Methods
- Surgically resected nonsmall cell lung carcinoma tissues, including 134 cases of squamous cell carcinoma (SqCC) and 99 cases of adenocarcinoma (ADC), were examined. In situ hybridization (ISH) for detecting episomal or integrated hrHPV DNA was performed using the INFORM HPV III Family 16 Probe (B) in the Ventana-validated assay. Anyplex II HPV28 detection kit based on real-time polymerase chain reaction was used for HPV DNA detection and genotyping.
-
Results
- All members of the study population were of Korean ethnicity. Episomal or integrated hrHPV DNA ISH analysis result was negative in all 233 cases. One SqCC of 89 samples (42 SqCCs and 47 ADCs) was positive for an hrHPV genotype by Anyplex II HPV28 detection kit.
-
Conclusion
- Our finding did not demonstrate hrHPV–etiologic correlation in primary lung SqCC and ADC in the Korean population.
- Human papillomavirus (HPV) is a double-stranded DNA virus and is a representative virus known to be an etiologic agent in both benign and malignant tumors. There are more than 100 different types of HPV, of which cancer-causing types are called “high-risk HPV (hrHPV).” The cancer-causing roles of hrHPV in uterine cervical carcinoma and oropharyngeal carcinoma are well established 1, 2, 3. Epidemiologic research, clinical research, and basic science have continued to clarify their relationship in head and neck cancer, anal cancer, esophageal cancer, and lung cancer 4, 5, 6. Based on a series of epidemiologic changes, a viral etiology has been theorized as playing a role in lung cancer initiation or evolution. Many studies have detected HPV DNA in lung cancer and the possible correlation between lung cancer and HPV has been suggested 6, 7, 8. However, inconsistent data regarding HPV DNA detection in lung cancer have been observed between studies. We considered that the heterogeneity of HPV DNA prevalence in lung cancer might be due to various HPV DNA detection methods as well as due to variations in geographical study origin and histological types of cancer. Usually, to detect HPV DNA, polymerase chain reaction (PCR) in DNA samples purified from tissue specimens and in situ hybridization (ISH) signal amplification in target tissues are used. A strong advantage of ISH is the direct visualization of HPV in either the episomal form or an integrated pattern within the nuclei of tumor cells in tissues.
- The aim of this study is to determine the prevalence of episomal or integrated hrHPV DNA in malignant cells of primary lung squamous cell carcinoma (SqCC) and adenocarcinoma (ADC) among Korean population, using the INFORM HPV III Family 16 Probe (B) in the Ventana-validated assay.
Introduction
- 2.1 Patients and tissue samples
- We obtained 233 formalin-fixed, paraffin-embedded (FFPE) lung samples from 233 patients who underwent surgical resection at Chungnam National University Hospital, Daejeon, South Korea, between 1995 and 2010. The tissues consisted of SqCC (n = 134) and 99 ADC (n = 99) samples. All samples were isolated from lobectomy or pneumonectomy specimens. Clinical records were reviewed. Of these, none of the 233 cases had any known history of head and neck primary cancer or uterine cervical cancer. None of the patients had received preoperative chemotherapy or radiotherapy. The lung cancer stages were determined according to the American Joint Committee on Cancer Staging System, Seventh Edition. Clinical data were provided by the National Biobank of Korea, Chungnam National University Hospital, Daejeon, South Korea. This study was approved by the Institutional Review Board of Chungnam National University Hospital.
- 2.2 Tissue microarray construction
- All cases were histologically reviewed by two pathologists (K.-H.K. and Y.-M.L.), and the two most representative areas of viable carcinoma tissue were selected and marked on the hematoxylin and eosin-stained slides. To create a tissue microarray (TMA), tissue columns (diameter, 3.0 mm) were punched from the original paraffin blocks and inserted into new recipient paraffin blocks (each containing 30 holes for tissue columns). Arrays were constructed using two 3-mm diameter cores per tumor.
- 2.3 High-risk HPV DNA ISH
- All TMA blocks and 20 whole-FFPE SqCC blocks were used for hrHPV DNA detection. ISH was performed using the Ventana INFORM HPV III Family 16 probe (B) kit (Catalog Number 800-4295; Ventana Medical System, Inc., Tucson, AZ, USA) according to the manufacturer's recommendations on the BenchMark, Automated Slide Staining System (Ventana Medical System, Inc.). The INFORM HPV III Family 16 probe (B) kit is composed of oligonucleotides designed to hybridize with hrHPV genotypes 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, and 66. The positive hrHPV signal demonstrated either episomal or integrated HPV. The episomal pattern appears as a large, homogeneous, globular navy-blue precipitate and the integrative pattern as a discrete, stippled navy blue nuclear pattern within the nuclei of neoplastic cells. Five hrHPV-positive cases of laryngeal SqCC were used as positive controls. Two pathologists independently reviewed the hrHPV ISH slides and a consensus was obtained after discussion.
- 2.4 Detection and genotyping of HPV DNA
- DNA was extracted from the whole-FFPE samples of 42 SqCCs and 47 ADCs. FFPE blocks were sectioned in six 7-μm sections. Total DNA was extracted using the G-DEX IIc Genomic DNA Extraction Kit according to manufacturer's instructions (iNtRON, Gyeonggi-do, South Korea).
- The Anyplex II HPV28 Detection kit (Seegene, Seoul, South Korea) identifies simultaneously 19 hrHPVs (16, 18, 26, 31, 33, 35, 39, 45, 51, 52, 53, 56, 58, 59, 66, 68, 69, 73, and 82) and nine low-risk HPVs (6, 11, 40, 42, 43, 44, 54, 61, and 70). Real-time PCRs were performed on a CFX96 Real-Time PCR System (Bio-Rad Laboratories, Hercules, CA, USA) with 20-mL reaction tubes containing HPV28 primer mix A, which recognizes 14 hrHPV types or mix B that identifies five hrHPV types and nine low-risk HPV types, Anyplex Master Mix (Seegene), and genomic DNA purified from specimen. For each run, positive controls of all HPV-type plasmid constructs were included along with a nontemplate control. Results were automatically analyzed in Seegene Viewer version 2.0 (Seegene, Seoul, South Korea) 9, 10.
Materials and methods
- We investigated 233 lung carcinoma samples. The demographics of the study patients are presented in Table 1. All members of the study population were of Korean ethnicity. hrHPV DNA ISH analysis was negative in all 233 TMA samples and 20 whole-FFPE SqCC samples (Figure 1).
- Eighty-nine whole-FFPE samples of 42 SqCCs and 47 ADCs were analyzed using the Anyplex II HPV28 Detection kit (Seegene) for detection and genotyping of HPV DNA. Only one SqCC of the total 89 samples (42 SqCCs and 47 ADCs) was positive for an HPV genotype, namely, hrHPV genotype 31. However, this positive case showed no signal amplification when subjected to HPV DNA ISH analysis using a whole-FFPE block sample.
Results
- No hrHPV DNA was demonstrated in our study, which included 134 SqCC cases and 99 ADC cases of the lung, in hrHPV DNA ISH analysis using Ventana INFORM HPV III Family 16 Probe (B). Other recent studies also failed to detect hrHPV DNA [Ventana INFORM HPV III Family 16 Probe (B)] and HPV E6/E7 messenger RNA (mRNA) using the RNAscope HPV kit (Advanced Cell Diagnostics Inc., Hayward, CA, USA) in primary lung SqCC and ADC 11, 12. Several other studies consistently have shown no presence of HPV16 E6/E7 mRNA by reverse transcription-PCR (RT-PCR) in primary lung SqCCs and ADCs 13, 14.
- The most commonly used method to detect HPV DNA is PCR in DNA purified from specimens. However, the sole finding of HPV DNA using PCR is not sufficient to prove the carcinogenic role of HPV in the development of cancer 12, 15. Compared with PCR, the HPV DNA ISH method permits visualization of HPV DNA signals using light microscopy to determine the prevalence of episomal or integrated HPV DNA in tissue specimens. To confirm that a tumor is caused by HPV, detecting the mRNA transcripts of HPV E6 and E7 viral oncogenes is necessary. Although detecting mRNA from FFPE tissue using RT-PCR is challenging, the RNAscope HPV kit (Advanced Cell Diagnostics Inc.) for RNA ISH probes complementary to E6/E7 mRNA permits direct visualization of viral transcripts in tissues [16]. Previous studies comparing HPV DNA ISH and RNA ISH assay found a high concordance rate in head and neck SqCC and nonsmall cell lung carcinoma 4, 12. Therefore, HPV DNA ISH is a feasible detection method in the clinical laboratory to prove that HPV is the etiologic factor for tumor development.
- An international pooled analysis showed that the highest prevalence of HPV16/18 DNA was observed in South and Central America, followed by Asia, North America, and Europe [7]. Our result does not support the previous studies showing a significantly higher detection rate of HPV in lung carcinoma in the Asian population than in the Australian, European, and North American populations 6, 7, 17, 18. The discordant results could be explained by the different detection methods.
- In conclusion, our results are in concordance with previous studies in which neither episomal nor integrated hrHPV DNA was detected in primary lung SqCC and ADC in hrHPV DNA ISH using Ventana INFORM HPV III Family 16 Probe (B). Thus, a causal relationship between hrHPV infection and carcinogenesis of primary lung SqCC and ADC was not demonstrated in the Korean population.
Discussion
- All authors have no conflicts of interest to declare.
Conflicts of interest
-
Acknowledgements
- This study was supported financially by the research fund of Chungnam National University.
Acknowledgments
- 1. Marur S., D'Souza G., Westra W.H.. HPV-associated head and neck cancer: a virus-related cancer epidemic. Lancet Oncol 11(8). 2010 Aug;781−789. PMID: 20451455.ArticlePubMed
- 2. Schiffman M., Castle P.E., Jeronimo J.. Human papillomavirus and cervical cancer. Lancet 370(9590). 2007 Sep 8;890−907. PMID: 17826171.Article
- 3. Parkin D.M., Bray F.. Chapter 2: the burden of HPV-related cancers. Vaccine 24(Suppl 3). 2006 Aug 31;S3/11–25.Article
- 4. Bishop J.A., Ma X.J., Wang H.. Detection of transcriptionally active high-risk HPV in patients with head and neck squamous cell carcinoma as visualized by a novel E6/E7 mRNA in situ hybridization method. Am J Surg Pathol 36(12). 2012 Dec;1874−1882. PMID: 23060353.ArticlePubMed
- 5. Bucchi D., Stracci F., Buonora N.. Human papillomavirus and gastrointestinal cancer: a review. World J Gastroenterol 22(33). 2016 Sep 7;7415−7430. PMID: 27672265.Article
- 6. Syrjanen K.. Detection of human papillomavirus in lung cancer: systematic review and meta-analysis. Anticancer Res 32(8). 2012 Aug;3235−3250. PMID: 22843898.PubMed
- 7. Ragin C., Obikoya-Malomo M., Kim S.. HPV-associated lung cancers: an international pooled analysis. Carcinogenesis 35(6). 2014 Jun;1267−1275. PMID: 24523449.ArticlePubMed
- 8. de Freitas A.C., Gurgel A.P., de Lima E.G.. Human papillomavirus and lung cancinogenesis: an overview. J Cancer Res Clin Oncol 142(12). 2016 Dec;2415−2427. PMID: 27357515.ArticlePubMed
- 9. Estrade C., Sahli R.. Comparison of Seegene Anyplex II HPV28 with the PGMY-CHUV assay for human papillomavirus genotyping. J Clin Microbiol 52(2). 2014 Feb;607−612. PMID: 24478495.ArticlePubMed
- 10. Kwon M.J., Roh K.H., Park H.. Comparison of the Anyplex II HPV28 assay with the Hybrid Capture 2 assay for the detection of HPV infection. J Clin Virol 59(4). 2014 Apr;246−249. PMID: 24568964.ArticlePubMed
- 11. Doxtader E.E., Katzenstein A.L.A.. The relationship between p16 expression and high-risk human papillomavirus infection in squamous cell carcinomas from sites other than uterine cervix: a study of 137 cases. Hum Pathol 43(3). 2012 Mar;327−332. PMID: 21840041.ArticlePubMed
- 12. Chang S.Y., Keeney M., Law M.. Detection of human papillomavirus in non-small cell carcinoma of the lung. Hum Pathol 46(11). 2015 Nov;1592−1597. PMID: 26342243.ArticlePubMed
- 13. Coissard C.J., Besson G., Polette M.C.. Prevalence of human papillomaviruses in lung carcinomas: a study of 218 cases. Mod Pathol 18(12). 2005 Dec;1606−1609. PMID: 16056245.ArticlePubMed
- 14. van Boerdonk R.A.A., Daniels J.M.A., Bloemena E.. High-risk human papillomavirus-positive lung cancer: molecular evidence for a pattern of pulmonary metastasis. J Thorac Oncol 8(6). 2013 Jun;711−718. PMID: 23571474.ArticlePubMed
- 15. Jordan R.C., Lingen M.W., Perez-Ordonez B.. Validation of methods for oropharyngeal cancer HPV status determination in US cooperative group trials. Am J Surg Pathol 36(7). 2012 Jul;945−954. PMID: 22743284.ArticlePubMed
- 16. Wang F., Flanagan J., Su N.. RNAscope: a novel in situ RNA analysis platform for formalin-fixed, paraffin-embedded tissues. J Mol Diagn 14(1). 2012 Jan;22−29. PMID: 22166544.ArticlePubMed
- 17. Chen Y.C., Chen J.H., Richard K.. Lung adenocarcinoma and human papillomavirus infection. Cancer 101(6). 2004 Sep 15;1428−1436. PMID: 15368331.Article
- 18. Cheng Y.W., Chiou H.L., Sheu G.T.. The association of human papillomavirus 16/18 infection with lung cancer among nonsmoking Taiwanese women. Cancer Res 61(7). 2001 Apr 1;2799−2803. PMID: 11306446.
References
Figure 1Squamous cell carcinoma. (A) A laryngeal squamous cell carcinoma positive for high-risk human papillomavirus (hrHPV). (B) A lung squamous cell carcinoma-negative in situ hybridization for hrHPV (Scale bar: 60 μm).
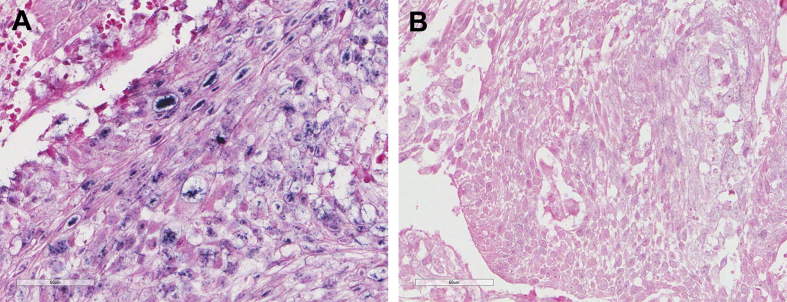

Table 1Demographics of the lung cancer patients (N = 233).
Figure & Data
References
Citations
Citations to this article as recorded by 

- HPV and lung cancer: A systematic review and meta‐analysis
Julia Karnosky, Wolfgang Dietmaier, Helge Knuettel, Viola Freigang, Myriam Koch, Franziska Koll, Florian Zeman, Christian Schulz
Cancer Reports.2021;[Epub] CrossRef - Presence and activity of HPV in primary lung cancer
Talita Helena Araujo de Oliveira, Carolina Medeiros do Amaral, Bianca de França São Marcos, Kamylla Conceição Gomes Nascimento, Ana Carine de Miranda Rios, Dafne Carolina Alves Quixabeira, Maria Tereza Cartaxo Muniz, Jacinto da Costa Silva Neto, Antonio C
Journal of Cancer Research and Clinical Oncology.2018; 144(12): 2367. CrossRef


 PubReader
PubReader Cite
Cite
