Articles
- Page Path
- HOME > Osong Public Health Res Perspect > Volume 7(5); 2016 > Article
-
Original Article
A Case–control Study on the Relationship betweenMycoplasma genitalium Infection in Women with Normal Pregnancy and Spontaneous Abortion using Polymerase Chain Reaction - Rashid Ramazanzadeha,b, Mazaher Khodabandehlooa,b, Fariba Farhadifarc, Samaneh Rouhia,b,d, Amjad Ahmadia,b, Shaho Menbarie, Fariba Fallahic, Reza Mirnejadf
-
Osong Public Health and Research Perspectives 2016;7(5):334-338.
DOI: https://doi.org/10.1016/j.phrp.2016.07.001
Published online: August 31, 2016
aCellular and Molecular Research Center, Kurdistan University of Medical Sciences, Sanandaj, Iran
bMicrobiology Department, Kurdistan University of Medical Sciences, Sanandaj, Iran
cSocial Determinants of Health Research Center, Kurdistan University of Medical Sciences, Sanandaj, Iran
dStudent Research Committee, Kurdistan University of Medical Sciences, Sanandaj, Iran
eDepartment of Pathology and Medical Laboratory Sciences, Kurdistan University of Medical Sciences, Sanandaj, Iran
fMolecular Biology Research Center, Baqiyatallah University of Medical Sciences, Tehran, Iran
- ∗Corresponding author. microbiol_sci@yahoo.com
© 2016 Korea Centers for Disease Control and Prevention. Published by Elsevier Korea LLC.
This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
Abstract
-
Objectives
- Mycoplasma genitalium infections are suggested as causes of a number of pathological outcomes in pregnant women. The aim of this study was to evaluate the frequency of M. genitalium infections among pregnant women and its association with spontaneous abortion.
-
Methods
- In this case–control study we included 109 women with spontaneous abortion with a gestational age of 10–20 weeks (patients), and 109 women with normal pregnancy with a gestational age of 20–37 weeks (controls) in Sanandaj, Iran. Using specific primers and extracted DNA from endocervical swabs, a polymerase chain reaction was conducted for the detection of M. genitalium infection in both groups.
-
Results
- The frequency of M. genitalium infection in patient and control groups was one (0.91%) and three (2.75%), respectively. In both control and patient groups using Fisher test, no association between mycoplasma infection and spontaneous abortion was seen.
-
Conclusion
- M. genitalium may be positive in the genital tract of some pregnant women but was not associated with spontaneous abortion. Further powerful studies with larger sample sizes are needed for the determination of a possible role of M. genitalium in pregnancy outcomes and spontaneous abortion.
- Spontaneous abortion prior to 20 weeks is a common adverse outcome of pregnancy, occurring in up to 15% of clinically recognized pregnancies. Usually spontaneous abortions occur in the first trimester due to phenotypic and/or chromosomal abnormalities, while environmental factors may have a greater impact on spontaneous abortion in later months of pregnancy [1]. Evidence suggests that maternal genitourinary and intrauterine infections have been proposed as an etiology for complications in pregnancy 2, 3. Mycoplasmas including Mycoplasma genitalium, Mycoplasma hominis, and Ureaplasma spp. are causes of sexually transmitted diseases (STDs) in men and women worldwide. About 80% of infected women and 50% of infected men may be asymptomatic [4]. M. genitalium and M. hominis are suggested as causes of a number of pathological outcomes in pregnant women. M. hominis was isolated from the amniotic fluid in 30% of women with intra-amniotic infection and was shown to be associated with preterm birth. M. genitalium is suggested to cause urethritis in men, mucopurulent cervicitis in women, and as an etiology of pelvic inflammatory disease independent of Chlamydia trachomatis and Neisseria gonorrhoeae 1, 5. In addition to cervicitis, it has been detected in confirmed salpingitis and endometritis.
- The most common pathway of intrauterine infection is vaginal and cervical infections and ascending route to amniotic cavity [6]. In addition, coinfection of mycoplasmas with other bacteria in the maternal genital tract during early gestation or preconception has been associated with spontaneous preterm birth, preterm premature rupture of the membranes, prematurity, spontaneous abortion, and perinatal morbidity and mortality 7, 8, 9. Although mycoplasmas have been associated with these morbidities, the exact role of the M. genitalium in the etiology of spontaneous abortion has not been determined because of the difficulty in culturing it from clinical samples. Thus, association between mycoplasma infection and pregnancy outcome seems to be more reliable due to the development of sensitive molecular diagnostic methods such as polymerase chain reaction (PCR) [10]. In a study in Brazil, Campos et al [11] using quantitative PCR in 302 women showed that M. hominis and M. genitalium were detected in 31.8% and 28.1% of samples, respectively. Ahmadi et al [12], in a case–control study in Iran on 109 women with spontaneous abortion (patients) and 109 women with normal pregnancy (controls) using PCR reported that in patients the rate of C. trachomatis infections was 22.9% and 11.9% in control. The association between C. trachomatis infection and spontaneous abortion had statistically significant. Hitti et al [13], in a case–control study in Peru using culture medium reported that in 661 cases with spontaneous preterm birth and 667 controls with normal delivery, M. genitalium was detected in 3% of them and was not a risk factor for preterm birth. Developed countries have established a national program for prenatal screening for infections in women, but in hospitals and clinics of Iran, there is such no national program [14]. The consequences of M. genitalium infection in pregnancy adverse outcome are unknown. The aim of this case–control study was to evaluate the frequency of M. genitalium infections among two groups of pregnant women (normal pregnancy and spontaneous abortion) and its association with spontaneous abortion.
Introduction
- 2.1 Participants
- This case–control and observational study was conducted in 2013. Pregnant women, attending the participating midwifery practices in the obstetrics and gynecology section or prenatal clinic and expected to deliver, were eligible for the study in Beasat Hospital, Sanandaj, Iran. We included 109 women (age, 19–43 years) with spontaneous abortion with a gestational age of 10–20 weeks (patients), and 109 (aged 19–43 years) women with normal pregnancy with a gestational age of 20–37 weeks (controls). Both groups included 26 women older than 25 years and 83 women younger than 25 years. Women expressed their informed consent to participate in this study. Demographic information such as age, education status, place of residence, occupation, and obstetrical data such as the number of childbirths, miscarriage, premature delivery, genital infection, urinary infection, the gestational age, and contraceptive methods before pregnancy were asked in a questionnaire and by a gynecologist. Inclusion criteria were having sexual activity and no use of antibiotics 2 weeks before sampling. Exclusion criteria were immunodeficiency, persons with chronic diseases (diabetes, endocrine disorders, and hypertension), recurrent miscarriage due to anatomic injury, and positive tests of first and second screening tests. In addition to asking when the 1st day of last menstruation was, ultrasound scan tests were done to estimate gestational age. To eliminate the threat of chromosomal abnormalities and probability of a genetic miscarriage, fetal health assessment tests were done at 11–13 weeks of gestation, including nuchal translucency, double tests such as pregnancy-associated plasma protein A and free β-human chorionic gonadotropin. In addition, at 15–17 weeks of gestation the confirmatory triple tests were performed for elimination of probable neural tube and chromosomal anomalies. After the matching of the two groups (by gestational age and place of residence) and completing the questionnaire, participants were asked for two endocervical secretion samples using a cotton swap. Samples were then immediately taken into sterile 15-mL falcon tubes containing 5 mL of phosphate-buffered saline and placed at −70°C until DNA extraction. This study had the written consent of all its participants in both groups and was approved by the Ethics Committee of the Kurdistan University of Medical Sciences.
- 2.2 DNA extraction
- Tubes containing cervical swab specimens were transported to the laboratory's compliance with the cold chain. Tubes were centrifuged at 6,000 rpm for 30 minutes. Then, the supernatant was discarded and the sediment was poured into the 1.5-mL microtube. The sediment was used for DNA extraction. DNA was extracted from sediments using a DNA extraction kit (High pure PCR Template Preparation; Roche, Germany) according to the manufacturer's instruction. After DNA extraction, to avoid degradation, the sample was split into separate 0.2-mL microtubes and maintained at −70°C until PCR diagnostic test.
- 2.3 PCR test for M. genitalium detection
- We designed specific primers for the adhesion protein gene of M. genitalium, genome sequence deposited in GeneBank. The M. genitalium primer sequences were: Forward: 5′-AGT TGA TGA AAC CTT AAC CCC TTG G-3′ and Reverse: 5′-CCG TTG AGG GGT TTT CCA TTT TTG C-3′. The length of the M. genitalium PCR product was 282 bp. Using designed primers and extracted DNA from cervical specimens, PCR was conducted. PCR reaction was done in a total volume of 25 μL of PCR master mix (CinnaGen, Tehran, Iran). The PCR amplification program in a Thermocycler (Eppendorf, Hamburg, Germany) was: initial denaturation 94°C for 10 minutes, followed by 30 cycles of denaturation at 94°C for 1 minute, annealing at 64°C for 30 seconds, and extension at 72°C for 1 minute, and final extension at 72°C for 10 minutes. PCR products were separated by gel electrophoresis apparatus (GE-100, Shanghai Beiyi Bioequip Information Co., Ltd 906 Lianye Building, No. 299 South Wulumuqi Road, Shanghai, 200031, P.R. China) in 1.5% gel agarose, stained by ethidium bromide, and visualized by UV light and then photographed. The PCR positive control was DNA extracted from isolated M. genitalium.
- 2.4 Statistical analysis
- The data were entered into SPSS 16.0 statistical software (SPSS, Chicago, IL, USA), and analyzed. Results were summarized and shown as tables and percentages. Fisher test was used for comparison of qualitative variables.
Material and methods
- The prevalence of M. genitalium infection was one (0.91%) in the patient group (spontaneous abortion) and three (2.75%) in the control group (normal pregnancy; Table 1). The age of the women ranged from 19 years to 43 years (29.6 ± 5.9 years) in the patient group and from 19 years to 42 years (27.8 ± 4.87 years) in the control group. Parity was two in the patients and control groups and both groups had two children (average number). Urinary and vaginal coinfections in the patient and control groups were observed in two and zero individuals, respectively (Table 1). The frequency of urinary tract infection in the husbands of patients was two, but zero the in control group. The frequency of smoking was three women in the patient group and three in the control group, and the frequency of alcohol consumption was zero in both groups. Demographic information and other patient information is presented in Table 1. A representative stained gel following PCR assay is shown in Figure 1. Using Fisher test, the association between M. genitalium infection and spontaneous abortion was found to be nonsignificant (odds ratio, 0.32; confidence interval, 0.03–0.19; p = 0.62).
Results
- Available data on the role of M. genitalium, infections in pregnancy adverse outcomes is increasing, but its role in spontaneous abortion is controversial. Dehon and McGowin [15], using real-time quantitative PCR reported that in 473 women with a low risk for acquiring STDs, the prevalence rate of M. genitalium was 1.5%. In our study, in patient and control groups, 10.09% and 4.58% of women had vaginal infections, respectively. Studies showed that M. genitalium is a common causative agent of STDs in men and women and is a reason for infertility among infected people [16]. In Iran, Amirmozafari et al [17] reported that in 210 genital swabs taken from 210 patients, mycoplasma strains were isolated from 39.5% of patients using a selective mycoplasma isolation media, but using PCR 57.1% of patient samples were found to be positive for mycoplasmas. In our study, PCR showed that the total prevalence of M. genitalium infection was four (3.66%) in patient and control groups. PCR is highly sensitive because genus specific primers are used for diagnosis of genital mycoplasmas in comparison with culture [17]. In Cuba, Mondeja et al [18] using a culture method in Vero cell suspensions, 16S ribosomal RNA, and MgPa1–3 PCR showed that all 11 isolates that were detected as M. genitalium with the culture method were also positive for M. genitalium with PCR [18]. Usually, PCR is used to detect M. genitalium infections, because it has proved to be more sensitive, specific, and rapid than the conventional microbiology methods 8, 19. For example, detection of M. genitalium by the culture method requires up to 8 weeks 9, 20.
- A study reported M. genitalium infections in symptomatic and asymptomatic men referred to the Urology Clinic in Tehran, Iran. The prevalence of M. genitalium was 12% and 2% in symptomatic and asymptomatic men, respectively, and M. genitalium infection was associated with dysuria, testis inflammation, pelvic pain, and low educational level [4]. In Mirnejad et al's [20] study, genital swabs were taken from 210 women in Tehran, Iran, and, using PCR the frequency of M. genitalium was 3.3%. The prevalence of C. trachomatis and M. genitalium in pregnant women of Sabzevar (north-east of Iran) in Haghighi Hasanabad et al's [9] study was determined in 196 urine specimens by duplex PCR. A total of 31 (15.81%) specimens were positive, with C. trachomatis in 27 (13.7%), M. genitalium in two (1.02%), and coinfection with both in two (1.02%) specimens. Also, significant correlation was found between preterm labor and infection [9]. In our results, prevalence of M. genitalium in patient and control groups were 0.91% and 2.75%, respectively. The results of M. genitalium prevalence in the aforementioned studies are nearly similar to our study. Differences of the results in some studies may be due to the type of population (pregnant vs. nonpregnant), sample types (urine vs. swab), date of study (recent vs. past), as well as the detection methodologies 1, 21, 22.
- Among cervical samples from 1,014 women in a case–control study, 6.2% were infected with M. genitalium in Guinea-Bissau using PCR assay; M. genitalium did not have a deleterious impact on the outcome of pregnancy [21]. In a prospective cohort study of 877 women with singleton pregnancies at < 11 weeks of gestation M. genitalium was examined in vaginal swabs by PCR in Japan. M. genitalium were detected in 0.8%, of the women. No significant association was found between preterm labor and M. genitalium [5], which is compatible with our findings: there was no association between M. genitalium infection and spontaneous abortion in our results. The lack of association in our study may be explained by health promotion in the population, and failure to identify potential confounding factors. However, Capoccia et al [8] showed that the role of Mycoplasma spp. and U. urealyticum in adverse pregnancy outcomes is increasing. In our study, history of vaginal infection in the patient group was somewhat more than control group, but this was not statistically significant. The presence of this microorganism in the vaginal flora may be insufficient to cause pathological problems, but combined with other factors such as other bacterial vaginosis or cervical incompetence may also induce spontaneous abortion. However, a screening program is important in sexually active women and needed as part of case-finding strategies and treatment. Whether detection and antimicrobial therapy of mycoplasma in pregnant women can effectively reduce the incidence of unwanted pregnancy outcomes needs further investigation.
- In conclusion, our study showed that M. genitalium may be positive in the genital tract of pregnant women but was not associated with spontaneous abortion. The low prevalence of M. genitalium and no association with pregnancy adverse outcome suggests that is unlikely to be an important risk factor in healthy women. Further powerful studies with larger sample sizes are needed for determination of possible role of M. genitalium in reproductive, pregnancy outcomes. Such studies should detect M. genitalium in cervical or vaginal specimens from women prior to pregnancy or early in gestation.
Discussion
- All authors have no conflicts of interest to declare.
Conflicts of interest
-
Acknowledgements
- This is part of Amjad Ahmadi's MSc in Medical Microbiology thesis. The authors wish to extend their gratitude to the Research Deputy of the Kurdistan University of Medical Sciences for financial support.
Acknowledgments
- 1. Short V.L., Jensen J.S., Nelson D.B.. Mycoplasma genitalium among young, urban pregnant women. Infect Dis Obstet Gynecol 2010:2010 Feb;1−8.ArticlePDF
- 2. Nigro G., Mazzocco M., Mattia E.. Role of the infections in recurrent spontaneous abortion. J Matern Fetal Neonatal Med 24(8). 2011 Aug;983−989. PMID: 21261443.ArticlePubMed
- 3. Petit E., Abergel A., Dedet B.. The role of infection in preterm birth. J Gynecol Obstet Biol Reprod (Paris) 41(1). 2012 Feb;14−25. [in French]. PMID: 22192232.ArticlePubMed
- 4. Yeganeh O., Jeddi-Tehrani M., Yaghmaie F.. A survey on the prevalence of Chlamydia trachomatis and Mycoplasma genitalium infections in symptomatic and asymptomatic men referring to urology clinic of Labbafinejad Hospital, Tehran, Iran. Iran Red Crescent Med J 15(4). 2013 Apr;340−344. PMID: 24083010.ArticlePubMed
- 5. Kataoka S., Yamada T., Chou K.. Association between preterm birth and vaginal colonization by mycoplasmas in early pregnancy. J Clin Microbiol 44(1). 2006 Jan;51−55. PMID: 16390947.ArticlePubMed
- 6. Choi S.J., Park S.D., Jang I.H.. The prevalence of vaginal microorganisms in pregnant women with preterm labor and preterm birth. Ann Lab Med 32(3). 2012 Apr;194−200. PMID: 22563554.ArticlePubMed
- 7. Kwak D.W., Hwang H.S., Kwon J.Y.. Co-infection with vaginal Ureaplasma urealyticum and Mycoplasma hominis increases adverse pregnancy outcomes in patients with preterm labor or preterm premature rupture of membranes. J Matern Fetal Neonatal Med 27(4). 2014 Jul;333−337. PMID: 23796000.ArticlePubMed
- 8. Capoccia R., Greub G., Baud D.. Ureaplasma urealyticum, Mycoplasma hominis and adverse pregnancy outcomes. Curr Opin Infect Dis 26(3). 2013 Jun;231−240. PMID: 23587772.ArticlePubMed
- 9. Haghighi Hasanabad M., Mohammadzadeh M., Bahador A.. Prevalence of Chlamydia trachomatis and Mycoplasma genitalium in pregnant women of Sabzevar-Iran. Iran J Microbiol 3(3). 2011 Sep;123−128. PMID: 22347594.PubMed
- 10. Wilkowska-Trojniel M., Zdrodowska-Stefanow B., Ostaszewska-Puchalska I.. The influence of Chlamydia trachomatis infection on spontaneous abortions. Adv Med Sci 54(1). 2009 Mar;86−90. PMID: 19403438.ArticlePubMed
- 11. Campos G.B., Lobão T.N., Selis N.N.. Prevalence of Mycoplasma genitalium and Mycoplasma hominis in urogenital tract of Brazilian women. BMC Infect Dis 15:2015 Feb;60PMID: 25886914.ArticlePubMed
- 12. Ahmadi A., Khodabandehloo M., Ramazanzadeh R.. The relationship between Chlamydia trachomatis genital infection and spontaneous abortion. J Reprod Infertil 17(2). 2016 Mar;110−116. PMID: 27141466.PubMed
- 13. Hitti J., Garcia P., Totten P.. Correlates of cervical Mycoplasma genitalium and risk of preterm birth among peruvian women. Sex Transm Dis 37(2). 2010 Feb;81−85. PMID: 20051932.ArticlePubMed
- 14. Fatholahzadeh B., Bahador A., Haghighi Hasanabad M.. Comparative screening of Chlamydia trachomatis infection in women population in Tehran, Iran. Iran Red Crescent Med J 14(5). 2012 May;289−293. PMID: 22829988.PubMed
- 15. Dehon P.M., McGowin C.L.. Mycoplasma genitalium infection is associated with microscopic signs of cervical inflammation in liquid cytology specimens. J Clin Microbiol 52(7). 2014 Jul;2398−2405. PMID: 24759719.ArticlePubMed
- 16. Mohseni Moghadam N., Kheirkhah B., Mirshekari T.. Isolation and molecular identification of Mycoplasma genitalium from the secretion of genital tract in infertile male and female. Iran J Reprod Med 12(9). 2014 Sep;601−608. PMID: 25469132.PubMed
- 17. Amirmozafari N., Mirnejad R., Kazemi B.. Comparison of polymerase chain reaction and culture for detection of genital mycoplasma in clinical samples from patients with genital infections. Saudi Med J 30(11). 2009 Nov;1401−1405. PMID: 19882050.PubMed
- 18. Mondeja B.A., Jensen J.S., Rodriguez I.. Isolation of Mycoplasma genitalium from patients with urogenital infections: first report from the Latin-American region. New Microbes New Infect 1(2). 2013 Nov;22−26. PMID: 25356322.ArticlePubMed
- 19. Perni S.C., Vardhana S., Korneeva I.. Mycoplasma hominis and Ureaplasma urealyticum in midtrimester amniotic fluid: association with amniotic fluid cytokine levels and pregnancy outcome. Am J Obstet Gynecol 191(4). 2004 Oct;1382−1386. PMID: 15507969.ArticlePubMed
- 20. Mirnejad R., Amirmozafari N., Kazemi B.. Simultaneous and rapid differential diagnosis of Mycoplasma genitalium and Ureaplasma urealyticum based on a polymerase chain reaction-restriction fragment length polymorphism. Indian J Med Microbiol 29(1). 2011 Jan–Mar;33−36. PMID: 21304192.ArticlePubMed
- 21. Labbe A.C., Frost E., Deslandes S.. Mycoplasma genitalium is not associated with adverse outcomes of pregnancy in Guinea-Bissau. Sex Transm Infect 78(4). 2002 Aug;289−291. PMID: 12181470.ArticlePubMed
- 22. Oakeshott P., Hay P., Taylor-Robinson D.. Prevalence of Mycoplasma genitalium in early pregnancy and relationship between its presence and pregnancy outcome. BJOG 111(12). 2004 Dec;1464−1467. PMID: 15663138.ArticlePubMed
References
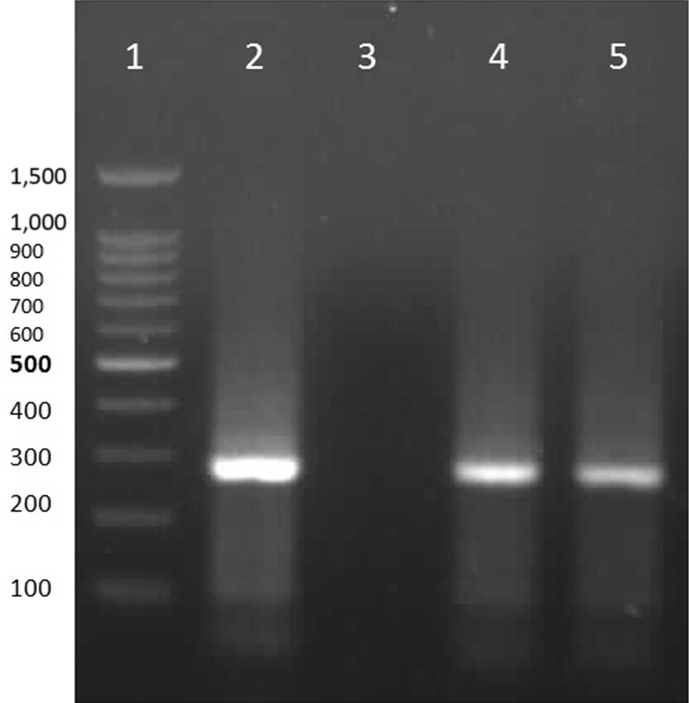
Figure & Data
References
Citations

- The association between genital mycoplasma infection and spontaneous abortion: A systematic review and meta-analysis
Wei Chen, Shimin Xiong, Xubo Shen, Ting Qiu, Caidie He, Songlin An, Yuanzhong Zhou
Reproductive Toxicology.2023; 116: 108334. CrossRef - Latest Advances in Laboratory Detection of Mycoplasma genitalium
Ken B. Waites, Donna M. Crabb, Amy E. Ratliff, William M. Geisler, T. Prescott Atkinson, Li Xiao, Romney M. Humphries
Journal of Clinical Microbiology.2023;[Epub] CrossRef - Weighing Potential Benefits and Harms of Mycoplasma genitalium Testing and Treatment Approaches
Lisa E. Manhart, William M. Geisler, Catriona S. Bradshaw, Jørgen S. Jensen, David H. Martin
Emerging Infectious Diseases.2022;[Epub] CrossRef - The Associations of Genital Mycoplasmas with Female Infertility and Adverse Pregnancy Outcomes: a Systematic Review and Meta-analysis
Caifeng Ma, Jikun Du, Yuhong Dou, Rui Chen, Yuxia Li, Lijun Zhao, Helu Liu, Kebin Zhang
Reproductive Sciences.2021; 28(11): 3013. CrossRef - Mollicutes antibiotic resistance profile and presence of genital abnormalities in couples attending an infertility clinic
Brenda Maldonado-Arriaga, Noé Escobar-Escamilla, Juan Carlos Pérez-Razo, Sofia Lizeth Alcaráz-Estrada, Ignacio Flores-Sánchez, Daniel Moreno-García, Rebeca Pérez-Cabeza de Vaca, Paul Mondragón-Terán, Jonathan Shaw, Cecilia Hernandez-Cortez, Graciela Castr
Journal of International Medical Research.2020; 48(1): 030006051982894. CrossRef - Association of spontaneous abortion and Ureaplasma parvum detected in placental tissue
C. N. T. Oliveira, M. T. S. Oliveira, H. B. M. Oliveira, L. S. C. Silva, R. S. Freire, M. N. Santos Júnior, M. V. Oliveira, J. Timenetsky, G. B. Campos, L. M. Marques
Epidemiology and Infection.2020;[Epub] CrossRef - Epidemiology of genital infections caused by Mycoplasma hominis, M. genitalium and Ureaplasma urealyticum in Iran; a systematic review and meta-analysis study (2000–2019)
Khadijeh Moridi, Mohammad Hemmaty, Amir Azimian, Mohammad Hosein Fallah, Hamid Khaneghahi Abyaneh, Kiarash Ghazvini
BMC Public Health.2020;[Epub] CrossRef - Prevalence and incidence of Mycoplasma genitalium in a cohort of HIV-infected and HIV-uninfected pregnant women in Cape Town, South Africa
Carolyn P Smullin, Hunter Green, Remco Peters, Dorothy Nyemba, Yamkela Qayiya, Landon Myer, Jeffrey Klausner, Dvora Joseph Davey
Sexually Transmitted Infections.2020; 96(7): 501. CrossRef - Frequency of Chlamydia trachomatis, Mycoplasma genitalium, and Ureaplasma urealyticum Isolated From Vaginal Samples of Women in Kerman, Iran
Zahra Zahirnia, Shahla Mansouri, Fereshteh Saffari, Ghazal Mansouri
Archives of Clinical Infectious Diseases.2018;[Epub] CrossRef - High Prevalence of Leptotrichia amnionii, Atopobium vaginae, Sneathia sanguinegens, and Factor 1 Microbes and Association of Spontaneous Abortion among Korean Women
Sang Soo Seo, Selvaraj Arokiyaraj, Mi Kyung Kim, Hea Young Oh, Minji Kwon, Ji Sook Kong, Moon Kyung Shin, Ye Lee Yu, Jae Kwan Lee
BioMed Research International.2017; 2017: 1. CrossRef


 PubReader
PubReader Cite
Cite
