Articles
- Page Path
- HOME > Osong Public Health Res Perspect > Volume 5(6); 2014 > Article
-
Original Article
Rapid DNA Extraction from Dried Blood Spots on Filter Paper: Potential Applications in Biobanking - Eun-Hye Choia, Sang Kwang Leea, Chunhwa Ihma,b, Young-Hak Sohnb
-
Osong Public Health and Research Perspectives 2014;5(6):351-357.
DOI: https://doi.org/10.1016/j.phrp.2014.09.005
Published online: November 1, 2014
aEulji Medi-Bio Research Institute (EMBRI), Daejeon, Korea
bDepartment of Laboratory Medicine, Eulji University Hospital, Daejeon, Korea
- ∗Corresponding author. medsohn@eulji.ac.kr
• Received: August 8, 2014 • Revised: September 14, 2014 • Accepted: September 29, 2014
© 2014 Published by Elsevier B.V. on behalf of Korea Centers for Disease Control and Prevention.
This is an Open Access article distributed under the terms of the CC-BY-NC License (http://creativecommons.org/licenses/by-nc/3.0).
Abstract
-
Objectives
- Dried blood spot (DBS) technology is a microsampling alternative to traditional plasma or serum sampling for pharmaco- or toxicokinetic evaluation. DBS technology has been applied to diagnostic screening in drug discovery, nonclinical, and clinical settings. We have developed an improved elution protocol involving boiling of blood spots dried on Whatman filter paper.
-
Methods
- The purpose of this study was to compare the quality, purity, and quantity of DNA isolated from frozen blood samples and DBSs. We optimized a method for extraction and estimation of DNA from blood spots dried on filter paper (3-mm FTA card). A single DBS containing 40 μL blood was used.
-
Results
- DNA was efficiently extracted in phosphate-buffered saline (PBS) or Tris-EDTA (TE) buffer by incubation at 37°C overnight. DNA was stable in DBSs that were stored at room temperature or frozen. The housekeeping genes GAPDH and beta-actin were used as positive standards for polymerase chain reaction (PCR) validation of general diagnostic screening.
-
Conclusion
- Our simple and convenient DBS storage and extraction methods are suitable for diagnostic screening by using very small volumes of blood collected on filter paper, and can be used in biobanks for blood sample storage.
- Biological samples collected in biobanks constitute a resource with significant research potential, but may also be subject to various ethical and deontological conflicts. Moreover, many samples now stored in biobanks were collected many years earlier, under nonstandardized conditions and in contexts where awareness of the need to meticulously handle information and obtain consent was not yet widespread. The problems this presents for the use of samples has been noted worldwide and has been addressed in different ways [1].
- In 2010, the Italian Joint Group CNB-CNBBSV (National Committee of Bioethics–National Committee for Biosecurity Biotechnologies and Life Sciences) published recommendations for the storage and use of dried blood spots (DBSs), and the development of a national network of regional newborn screening repositories for the collection of residual DBSs [2].
- Collection of blood samples on filter paper was first performed >50 years earlier [1]. This simple collection and storage method has broadly been adopted in diagnostic screening [3], drug monitoring [4], and genetic analysis [5], and is particularly suitable for molecular epidemiologic studies in remote tropical areas, where transport and storage conditions are often suboptimal.
- The DBSs used in this study were obtained by finger or heel pricks, and were spotted onto filter paper. The analysis of human blood dates back to 1963, when Dr Robert Guthrie used DBS specimens for phenylalanine analysis to identify newborns with phenylketonuria. This novel approach to blood collection facilitated population screening of newborns and other clinical testing applications [6].
- DBSs can easily be collected by a heel or finger prick without the need for venipuncture; they can be stored at room temperature and easily transported to the testing laboratory with little biohazard risk. The use of stored DBS samples combined with clinical information from medical registries constitutes an ideal resource for large population studies of patients under a given age without selection bias. In addition, DBSs offer several advantages in areas that lack appropriate infrastructure for processing blood and a cold chain for the transportation and storage of blood products [7].
- Generally, the use of neonatal DBSs is based on the removal of blood spots using punches, typically 3–6 mm in diameter, followed by physical or biochemical analysis of serum analytes released from the punched-out material by soaking it in alcohol or water [8]. More recently, DBSs have been used to screen for heritable traits at the DNA level; typically, they have been used for traits such as cystic fibrosis, thalassemia, and other traits that are readily assessed by polymerase chain reaction (PCR).
- The use of DBSs has expanded from their modest beginning in the diagnostic arena for neonatal screening [3] to drug development, including discovery research [9,10], nonclinical and clinical toxico- and pharmacokinetic evaluations, and therapeutic drug monitoring [11–13]. This is due to the multifold advantages of DBS with respect to scientific, practical, ethical, and cost-effective aspects in nonclinical and clinical settings [14–17].
- DBSs represent a valuable and comprehensive genetic repository because genomic DNA can be isolated from blood samples processed on these filter papers for use in PCR [18]. In addition, direct PCR amplification from DBSs that have been punched out has been reported [19].
- We analyzed whole blood and buffy coats in comparison with DBSs, in terms of storage temperature, buffer stability, and DNA yield. We have presented a storage and extraction method for DBSs on filter paper, with PCR verification. We have developed a reliable system for good blood storage practice, blood sample preparation and processing, conditions, and acceptance criteria for DBS applications.
Introduction
- 2.1 Collection and storage of blood specimens
- Blood samples were collected in EDTA tubes from unaffected individuals tested at the Eulji University Hospital, Daejeon, Korea. All blood samples were obtained from young adults (20–40 years) as whole blood in EDTA and as DBS samples.
- DBS samples (40 μL each) were spotted on Whatman FTA cards (Whatman Inc., Brentford, UK) and were dried for 24 hours at room temperature. They were stored at room temperature or at −80°C until analysis, and then kept in separate clean zipper bags for 2, 4 weeks and 3 months. Liquid, EDTA-treated blood samples were also stored at 80°C for 2, 4 weeks and 3 months.
- 2.2 DNA extraction from DBSs
- For single-stranded DNA (ssDNA) extraction, DBSs on the cards were cut into small pieces with scissors and transferred into 1.5-mL microtubes to which 500 μL dH2O had been added; the tubes were kept at room temperature for 5 minutes. The dH2O was discarded, and the procedure was repeated twice. The filter papers were soaked in 500 μL extraction buffer [phosphate-buffered saline (PBS) or 10 mM Tris-EDTA (TE)].
- For lysis, red blood cell lysis buffer (100 μL) was added to the filter papers, which were then incubated with 10 μL (1 mg/mL) proteinase K at 37°C overnight. After blood cell lysis, the filter papers were pressed against the bottom of the tube several times with a clean pipette tip [20]. For increased extraction efficiency, cell lysates were incubated at 95°C for 15 minutes. Filter papers were removed after brief centrifugation (2–3 seconds), and the DNA-containing supernatant was stored at −80°C until analysis.
- For double-stranded DNA (dsDNA) extraction, each filter paper sample that had been punched out was soaked in 40 μL Tris-EDTA buffer or PBS, and incubated at 37°C overnight. Genomic DNA was isolated from blood using the DNeasy Blood and Tissue kit (Qiagen, Valencia, CA, USA) as per the manufacturer's instructions.
- 2.3 DNA concentration
- DNA concentrations were determined by measuring absorbance at 260 nm (A260), and assuming that an A260 of 1.0 is equal to 50 ng/μL pure dsDNA or 35 ng/μL pure ssDNA. Concentrations of extracted DNA were measured using a SmartSpec plus spectrophotometer (Bio-Rad, Hercules, CA, USA).
- 2.4 Polymerase chain reaction
- The levels of the GAPDH and β-actin transcripts were used to verify the quality of the extracted DNA. For GAPDH PCR, the PCR reaction mixture contained 3 μL extracted DNA, 10 pmol of each primer, and Bioneer PCR premix in a final volume of 20 μL. Thermal cycling was conducted in a CFX96 Touch system (Bio-Rad), with the following PCR program for GAPDH: initial denaturation of 8 minutes at 94°C; followed by 40 cycles of 94°C for 1 minute, 65°C for 1 minute, and 72°C for 1 minute; concluding with 7 minutes at 72°C and 1 minute at 4°C. A similar PCR program was used for β-actin PCR, except that the annealing temperature was 64°C. The primer sequences for GAPDH and β-actin are shown in Table 1 [21].
- The PCR products were analyzed by electrophoresis on 2% RedSafe (Koma)-stained agarose gels in Tris-acetate-EDTA (TAE) buffer. After electrophoresis, the digital images were analyzed with a ChemiDoc MP imaging system (Bio-Rad).
Materials and methods
- 3.1 Quantitation of DNA from filter paper and frozen-liquid samples
- The recovery ratio of dsDNA was measured as described in the “Materials and Methods” section. The dsDNA extracted from DBSs and liquid samples was stored at room temperature or −80°C for 3 months (Figure 1).
- As shown in Figure 2, the recovery ratios of frozen-liquid and filter paper samples were compared. The dsDNA was reduced by approximately 68% in the whole blood and 40% in the buffy coat. The amount of DNA extracted from the frozen whole blood was approximately 39% higher than that extracted from the frozen buffy coat. The amount of DNA extracted from whole blood kept on filter paper at room temperature was approximately 11% higher than the yield from the buffy coat. The stability of DNA in samples collected on filter paper was higher than that for frozen-liquid samples. Furthermore, in a comparison of samples stored in the same manner, the quantity of DNA extracted from whole-blood samples was approximately 39% higher than the yield from buffy-coat samples stored frozen and approximately 11% higher than that from samples stored on filter paper at room temperature.
- The ssDNA yields were measured following the procedure described in Figure 1. We determined that two storage methods were significantly higher extraction method of ssDNA quantity. The amount of ssDNA extracted from samples stored for 2 weeks was similar to that from samples stored for 4 weeks (Figure 3).
- The amount of DNA extracted from DBSs was >62% and 60% of that obtained from frozen-liquid samples (whole blood and buffy coats, respectively). The amounts of DNA extracted from frozen-liquid samples and filter paper samples at 2 weeks did not differ much from those extracted at 4 weeks; however, the yield from frozen-liquid samples was approximately 62% lower than that from filter paper samples after 2 weeks and approximately 60% lower after 4 weeks. The stability and yield of frozen-liquid samples were associated with storage temperature.
- DNA from DBSs stored at room temperature was of lower purity than that obtained from frozen-liquid samples because of impurities in the paper; however, the improved yield of the extracted ssDNA suggested greater stability than that obtained in the case of frozen-liquid samples.
- 3.2 Quantitation of ssDNA from DBS
- To measure the DNA yields from samples stored under different conditions, samples were separated into whole-blood and buffy-coat samples, spotted on filter papers, and stored at room temperature; subsequently, the extracted ssDNA yields were compared (Figure 4). The amount of ssDNA extracted from buffy-coat samples was approximately 65% higher than that extracted from whole-blood samples after 2 weeks and approximately 44% higher after 4 weeks. Moreover, the purity of ssDNA extracted from buffy-coat samples was higher than that extracted from whole-blood samples (data not shown).
- Buffy-coat sample yields were approximately three times higher than whole-blood sample yields after 2 weeks and approximately 2.4 times higher after 4 weeks; however, the difference decreased as the storage period increased. In the case of whole blood, there was little difference in ssDNA yield over time. However, the extraction yield from buffy coats decreased by approximately 20% after 2–4 weeks.
- 3.3 Yield of ssDNA from DBS at different storage temperatures and times
- As the filter paper storage method provides higher ssDNA yields than the frozen-liquid method at room temperature, we investigated how the storage temperature affected yields. As shown in Figure 5, ssDNA yields were compared between filter paper storage temperatures and times; there was little difference between the amounts of ssDNA extracted. However, there was a difference of 8% in the ssDNA yields between 2- and 4-week storage, while there was a 1% difference in ssDNA yields from samples stored at room temperature for the same period. Therefore, storage at room temperature was best for samples on filter paper.
- 3.4 Quantitation of ssDNA extracted from DBSs with different buffers
- Extraction yields with PBS and TE buffers were compared (Figure 6). Neither buffer produced much difference in yields for different sample storage times; however, the ssDNA yield in TE buffer was approximately 5% higher than that in PBS buffer. Therefore, TE buffer was more suitable than PBS for optimal ssDNA yield.
- 3.5 Validation by PCR
- To examine whether the extracted ssDNA could be used for PCR in medical and scientific research, we amplified GAPDH and β-actin from dsDNA or ssDNA extracted from frozen-liquid and filter paper samples.
- Nonquantitative PCR amplification of GAPDH and β-actin DNA was performed with (1) ssDNA from frozen-liquid samples, (2) dsDNA from filter paper, and (3) ssDNA from filter paper (Figure 7). The results showed that the DNA obtained from DBS extraction was of sufficient quality to be used for PCR analysis and that the yield was better than that obtained from frozen samples.
Results
- Compared with conventional plasma sampling, DBS requires much smaller blood volumes and simpler storage and transportation conditions, which makes it a very promising alternative sample-collection procedure. DBS sample preparation requires extraction of dried blood samples into a solution. This is a step unique to DBSs for large-molecule sample analysis and is crucial to the success of the method. For a given analyte, poor extraction recovery or poor correlation between the serum/plasma and DBS samples may indicate that further development of the method is necessary or that the analyte may not be a good candidate for the DBS method [22]. Therefore, sample-extraction conditions and recovery should be evaluated during method development and confirmed during method validation.
- DBS sampling is most commonly used in clinical laboratories to assess small-molecule analytes during screening of newborns for metabolic disorders [2]. Samples are prepared by spotting a small drop of blood directly onto the DBS collection card; only a portion of the DBS is removed for analysis. Ideally, the volume of blood transferred for the assay is defined by the diameter of the punch and, therefore, does not require precise measurement of the volume of blood that is spotted. However, there are several sources of variation unique to bloodspot sampling. For example, proper placement of the whole-blood sample on the filter paper is critical, and errors can be introduced if blood is blotted or smeared rather than drawn onto the filter paper by capillary action 16. We minimized this source of error by rigorous training of the personnel for the procedure and punching out of the entire blood spot. We focused on increasing the DNA extraction yield from blood specimens and maintaining high structural stability of DNA to retain the most information, with the intent of securing high-quality human specimens for future medical and scientific research. We established a method to obtain high-quality DNA for research by analyzing dsDNA and ssDNA yields after varying the storage method, temperature, and elution buffer, all of which influence DNA extraction.
- The importance of DNA recovery from Guthrie cards has been discussed at length in a recent comparative study by Sjöholm and colleagues [24]. They compared a number of commercially available kits and procedures for DNA recovery from Guthrie cards and demonstrated that only approximately 15–25% of the total DNA complement can be recovered. They measured DNA recovery from DBSs stored for up to 26 years and showed that standard 3-mm punches provide DNA yields (with the best available technology) of only approximately 30 ng per punch. However, they demonstrated that, in spite of the relatively low yields, the small amount of DNA obtained remains an excellent substrate for whole genome amplification and relatively complex multiplex single nucleotide polymorphism (SNP) analysis [24].
- In the current study, a comparison of frozen-liquid storage and DBSs showed that the quantity of dsDNA extracted using DNA preparation kits was higher after storage as frozen liquid. The mean dsDNA concentration from frozen-liquid samples was 87 ng/μL, with a standard deviation of 9.6 ng/μL. However, the mean dsDNA concentration obtained from the DBS samples was 23 ng/μL, with a standard deviation of 1.7 ng/μL.
- When economics are considered, the frozen-liquid method is limited by manpower and capital requirements. In this regard, DBSs stored at room temperature provide many economic benefits such as reduced cost and space requirements. However, genome-scale microarray analysis continues to require an input DNA concentration (250 ng) that is approximately 100 times greater than that required for simple PCR; the DNA must be double stranded and must contain intact molecules approximately five times longer than those required for most PCR reactions.
- Therefore, we tried to extract a large amount of DNA by optimization of an ssDNA extraction method for a customized DNA resource that can be used for PCR, which is widely used in DNA research. Extraction was performed after converting dsDNA to ssDNA by boiling the filter paper sample at 95°C, thereby releasing a large amount of DNA into the buffer. The mean ssDNA concentration obtained from the DBS samples was 24.5 μg/μL, with a standard deviation of 0.54 μg/μL.
- Similar results were obtained by Mas et al [23] in a study on DBSs stored on treated filter paper matrices such as Whatman FTA or IsoCode, wherein extraction was performed according to the manufacturer's instructions. In that study, approximately 25% recovery was obtained in a single extraction, yielding up to 150 ng of ssDNA in 200 μL solution, per 40 μL adult human blood input.
- The general recommendation is DBS sample storage at −20°C to prevent DNA degradation [24]. We compared DBS storage at room temperature versus that at −80°C. As shown in Figure 5, the concentration of ssDNA from DBS stored at −80°C was similar to the yield from DBS stored at room temperature, suggesting that our modified extraction method can be used for extraction of ssDNA from DBSs stored at room temperature.
- The ssDNA products were compared with dsDNA products for use in common PCR experiments (Figure 7). GAPDH and β-actin, human housekeeping genes, were collinearly expressed in the DBSs and frozen samples. Thus, DBSs may not be used in microarray technology, but may be used to analyze the quantity and quality of ssDNA.
- In this study, we analyzed the concentration of DNA extracted from DBS samples and frozen-liquid samples from the same healthy adults using a modified DNA extraction procedure and a commercial kit. The comparison revealed a slight advantage of the frozen-liquid samples over DBS samples with respect to dsDNA quantity, but the concentration of ssDNA was significantly greater in the DBS samples.
- The advantages of sampling and storing blood on filter paper for genetic analyses of human pathogens highlight the need for reliable, sensitive, and cost-effective DNA extraction methods. The denaturation-based DNA extraction method described in this report has shown superior results in comparison to standard methods and commercial kits for the extraction of archived blood samples. Our method is rapid, simple, and inexpensive, and confers a reduced risk of cross-contamination due to minimal sample manipulation. Using our DNA extraction method, archival DBS samples can provide sufficient DNA for clinical studies and biobanking.
Discussion
- Authors declare no conflicts of interest.
Conflicts of interest
-
Acknowledgements
- This work was supported by the Bio-Meditech Regional Innovation Center at Eulji University, under the Regional Innovation Center Program (Grant No. 2012-02-05) of the Ministry of Commerce, Industry, and Energy.
Acknowledgments
-
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article information
- 1. Petrini C., Olivieri A., Corbetta C.. Common criteria among States for storage and use of dried blood spot specimens after newborn screening. Ann Ist Super Sanita 48(2). 2012;119−121. PMID: 22751553.ArticlePubMed
- 2. Lang P.O., Govind S., Drame M.. Comparison of manual and automated DNA purification for measuring TREC in dried blood spot (DBS) samples with qPCR. J Immunol Methods 384(1–2). 2012 Oct;118−127. PMID: 22867745.ArticlePubMed
- 3. Guthrie R., Susi A.. A simple phenylalanine method for detecting phenylketonuria in large populations of newborn infants. Pediatrics 32:1963 Sep;338−343. PMID: 14063511.ArticlePubMedPDF
- 4. Lindstrom B., Ericsson O., Alvan G.. Determination of chloroquine and its desethyl metabolite in whole blood: an application for samples collected in capillary tubes and dried on filter paper. Ther Drug Monit 7(2). 1985;207−210. PMID: 4024215.ArticlePubMed
- 5. Prior T.W., Highsmith W.E. Jr., Friedman K.J.. A model for molecular screening of newborns: simultaneous detection of Duchenne/Becker muscular dystrophies and cystic fibrosis. Clin Chem 36(10). 1990 Oct;1756−1759. PMID: 2208650.ArticlePubMed
- 6. Wang C., Zhu H., Zhang W.. Second-tier test for quantification of underivatized amino acids in dry blood spot for metabolic diseases in newborn screening. Amino Acids 44(2). 2013 Feb;661−671. PMID: 22932943.ArticlePubMed
- 7. Hollegaard M.V., Grove J., Grauholm J.. Robustness of genome-wide scanning using archived dried blood spot samples as a DNA source. BMC Genet 12:2011 Jul;58−64. PMID: 21726430.ArticlePubMed
- 8. Wilcken B., Wiley V.. Newborn screening. Pathology 40:2008;104−115. PMID: 18203033.ArticlePubMed
- 9. Beaudette P., Bateman K.P.. Discovery stage pharmacokinetics using dried blood spots. J Chromatogr B Analyt Technol Biomed Life Sci 809(1). 2004 Sep;153−158.ArticlePubMed
- 10. Clark G.T., Haynes J.J., Bayliss M.A.. Utilization of DBS within drug discovery: development of a serial microsampling pharmacokinetic study in mice. Bioanalysis 2(8). 2010 Aug;1477−1488. PMID: 21083348.ArticlePubMed
- 11. Evans C.A.. Dried blood spot analysis: a paradigm shift. AAPS Newsmagazine 2010 April.
- 12. Edelbroek P.M., van der Heijden J., Stolk L.M.. Dried blood spot methods in therapeutic drug monitoring: methods, assays, and pitfalls. Ther Drug Monit 31(3). 2009 Jun;327−336. PMID: 19349929.ArticlePubMed
- 13. Spooner N., Lad R., Barfield M.. Dried blood spots as a sample collection technique for the determination of pharmacokinetics in clinical studies: considerations for the validation of a quantitative bioanalytical method. Anal Chem 81(4). 2009 Feb;1557−1563. PMID: 19154107.ArticlePubMed
- 14. Arnaud C.. Technology renews a basic approach: dried blood spots offer advantages, but also challenges for pharmaceutical analysis. Chem Eng News 89(3). 2011 Jan;13−17.Article
- 15. Corran P.H., Cook J., Lynch C.. Dried blood spots as a source of anti-malarial antibodies for epidemiological studies. Malar J 7:2008 Sep;195−206. PMID: 18826573.ArticlePubMed
- 16. McDade T.W., Williams S., Snodgrass J.J.. What a drop can do: dried blood spots as a minimally invasive method for integrating biomarkers into population-based research. Demography 44(4). 2007 Nov;899−925. PMID: 18232218.ArticlePubMed
- 17. Patel P., Mulla H., Tanna S.. Facilitating pharmacokinetic studies in children: a new use of dried blood spots. Arch Dis Child 95(6). 2010 Jun;484−487. PMID: 20501544.ArticlePubMed
- 18. Gupta B.P., Jayasuryan N., Jameel S.. Direct detection of hepatitis B virus from dried blood spots by polymerase chain reaction amplification. J Clin Microbiol 30(8). 1992 Aug;1913−1916. PMID: 1500493.ArticlePubMed
- 19. Huges D., Hurd C., Williamson L.M.. Genotyping for human platelet antigen-1 directly from dried blood spots on cards. Blood 88(8). 1996 Oct;3242−3243. PMID: 8874226.ArticlePubMed
- 20. Bereczky S., Martensson A., Gil J.P.. Short report: rapid DNA extraction from archive blood spots on filter paper for genotyping of Plasmodium falciparum. Am J Trop Med Hyg 72(3). 2005 Mar;249−251. PMID: 15772315.ArticlePubMed
- 21. Glare E.M., Divjak M., Bailey M.J.. beta-Actin and GAPDH housekeeping gene expression in asthmatic airways is variable and not suitable for normalising mRNA levels. Thorax 57(9). 2002 Sep;765−770. PMID: 12200519.ArticlePubMed
- 22. Burns D., Rajendran S., Wang J.. Validation feasibility of large molecules of differing modalities using dried blood spot samples. Poster presented at the 2011 AAPS—National Biotechnology Conference; San Francisco, CA. 2011.
- 23. Mas S., Crescenti A., Gasso P.. DNA cards: determinants of DNA yield and quality in collecting genetic samples for pharmacogenetic studies. Basic Clin Pharmacol Toxicol 101(2). 2007;132−137. PMID: 17651316.ArticlePubMed
- 24. Sjöholm M.I., Dillner J., Carlson J.. Assessing quality and functionality of DNA from fresh and archival dried blood spots and recommendations for quality control guidelines. Clin Chem 53(8). 2007 Aug;1401−1407. PMID: 17586590.ArticlePubMed
References
Figure 1Workflow diagram. dsDNA = double-stranded DNA; RBC = red blood cell; ssDNA = single-stranded DNA.
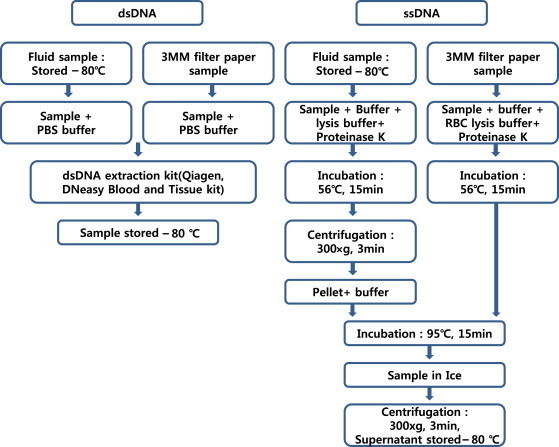

Figure 2DNA recovery from whole blood and Whatman FTA cards. BC = buffy coat in dried blood spots stored at room temperature; dsDNA = double-stranded DNA; WB = whole blood in dried blood spots stored at room temperature.


Figure 3Average DNA yield over time. In the figure, fluid represents whole blood stored at −80°C and paper represents Whatman FTA cards stored at room temperature. ssDNA = single-stranded DNA.
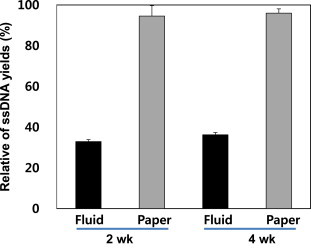

Figure 4DNA extracted by boiling dried blood spots on filter paper after incubation at 56°C. BC = buffy coat in dried blood spots stored at room temperature; ssDNA = single-stranded DNA; WB = whole blood in dried blood spots stored at room temperature.
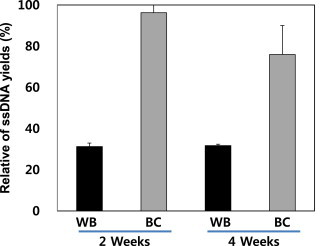

Figure 5Yield of ssDNA from dried blood spots stored at −80°C and room temperature. ssDNA = single-stranded DNA.
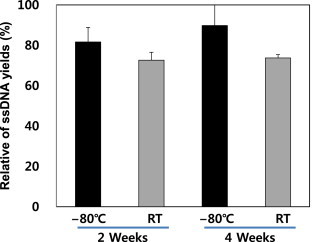

Figure 6Efficiency of ssDNA extraction in PBS and TE. PBS = phosphate-buffered saline; ssDNA = single-stranded DNA.
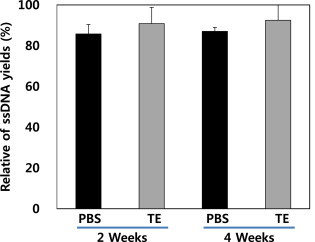

Figure 7PCR amplification of GAPDH and β-actin DNA. DNA was extracted from frozen-liquid samples and dried blood spots. Lanes 1–2 show dsDNA isolated from frozen whole blood and the buffy coat; lanes 3–4, dsDNA isolated from DBSs, with whole blood stored at room temperature; and lanes 5–6, ssDNA isolated from DBSs, with whole blood and the buffy coat stored at room temperature and extracted with the boiling method. BC = buffy coat in dried blood spots stored at room temperature; DBS = dried blood spot; dsDNA = double-stranded DNA; PCR = polymerase chain reaction; ssDNA = single-stranded DNA; WB = whole blood in dried blood spots stored at room temperature.
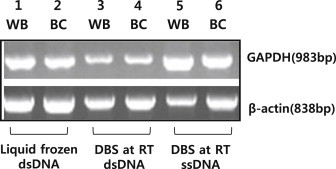

Figure & Data
References
Citations
Citations to this article as recorded by 

- Haptoglobin gene polymorphism and iron profile in sickle cell disease patients with inflammation in Yaounde, Cameroon
Romaric De Manfouo Tuono, Josué Louokdom Simo, Prosper Cabral Biapa Nya, Jean Paul Chedjou, Christian Bernard Kengne Fotsing, Bernard Claude Chetcha, Calvino Fomboh Tah, Claude Tagny Tayou, Wilfried Fon Mbatcham, Constant Anatole Pieme
Molecular Genetics & Genomic Medicine.2024;[Epub] CrossRef - CRISPR-Based Diagnostics: Challenges and Potential Solutions toward Point-of-Care Applications
Ahmed Ghouneimy, Ahmed Mahas, Tin Marsic, Rashid Aman, Magdy Mahfouz
ACS Synthetic Biology.2023; 12(1): 1. CrossRef - Effect of storage temperature and duration on direct PCR amplification of various feather types and DBS matrices
Maryam Aslam, Fatima Naeem, Rijaab Seher, Muhammad Zubair Shabbir, Wasim Shehzad, Muhammad Imran
Gene.2023; 854: 147116. CrossRef - Improvement of bovine pestiviral diagnosis by the development of a cost-effective method for detecting viral RNA in fresh specimens and samples spotted in filter papers
Paula M. Favaro, Ana Molineri, Maria J. Dus Santos, Luis F. Calvinho, Andrea Pecora
Revista Argentina de Microbiología.2023; 55(2): 167. CrossRef - A dual paper-based nucleic acid extraction method from blood in under ten minutes for point-of-care diagnostics
Kenny Malpartida-Cardenas, Jake Baum, Aubrey Cunnington, Pantelis Georgiou, Jesus Rodriguez-Manzano
The Analyst.2023; 148(13): 3036. CrossRef - Insight into Increased Recovery and Simplification of Genomic DNA Extraction Methods from Dried Blood Spots
Kiara Lee, Anubhav Tripathi
Biopreservation and Biobanking.2023;[Epub] CrossRef - Current Understanding of Dried Spots Platform for Blood Proteomics
Luis Perpétuo, Hugo Rocha, Rita Ferreira, Bruno Manadas, Francisco Amado, Sofia Guedes, Atef Mahmoud Mannaa, Jerome Vialaret, Christophe Hirtz, Rui Vitorino
Current Proteomics.2023; 20(2): 81. CrossRef - Comparative analysis of reagent kits for DNA extraction from dry blood stains
A. V. Sedykh, M. A. Saitgalina, Yu. V. Ostankova, A. A. Totolian
Medical Immunology (Russia).2023; 25(6): 1453. CrossRef - Large-scale Extraction of DNA by Using Salting-out Principle for Dried Blood Spots to Screen Multiple Mutations in GCDH Gene
Muntaj Shaik, A. Alladi, AB. Vedamurthy, KS. Devaraju, M. Kamate, TP. Kruthika-Vinod
Iranian Journal of Science and Technology, Transac.2022; 46(1): 33. CrossRef - Towards Next-Generation Sequencing (NGS)-Based Newborn Screening: A Technical Study to Prepare for the Challenges Ahead
Abigail Veldman, Mensiena B. G. Kiewiet, Margaretha Rebecca Heiner-Fokkema, Marcel R. Nelen, Richard J. Sinke, Birgit Sikkema-Raddatz, Els Voorhoeve, Dineke Westra, Martijn E. T. Dollé, Peter C. J. I. Schielen, Francjan J. van Spronsen
International Journal of Neonatal Screening.2022; 8(1): 17. CrossRef - Upregulated NOTCH Signaling in the Lens of Patients With Pseudoexfoliation Syndrome Compared With Pseudoexfoliation Glaucoma Suggests Protective Role
Zia S. Pradhan, Shivani Dixit, Lekshmi Krishna, Reshma Shetty, Sushma Tejwani, Harsha L. Rao, Rohit Shetty, Carroll A.B. Webers, Debashish Das
Journal of Glaucoma.2022; 31(3): e1. CrossRef - Electro-DBS: A Simple Method to Rapidly Extract Genomic DNA from Dried Blood Spots
Kiara Lee, John Murphy, Anubhav Tripathi
Analytical Chemistry.2022; 94(39): 13404. CrossRef - Population genomics of a predatory mammal reveals patterns of decline and impacts of exposure to toxic toads
Brenton von Takach, Louis Ranjard, Christopher P. Burridge, Skye F. Cameron, Teigan Cremona, Mark D. B. Eldridge, Diana O. Fisher, Stephen Frankenberg, Brydie M. Hill, Rosemary Hohnen, Chris J. Jolly, Ella Kelly, Anna J. MacDonald, Adnan Moussalli, Kym Ot
Molecular Ecology.2022; 31(21): 5468. CrossRef - A handmade DNA extraction kit using laundry powder; insights on simplicity, cost-efficiency, rapidity, safety and the quality of purified DNA
Reza Talebi, Ramin Seighalani, Saber Qanbari
Animal Biotechnology.2021; 32(3): 388. CrossRef - Molecular Investigation on Tick-Borne Hemoparasites and Coxiella burnetii in Dromedary Camels (Camelusdromedarius) in Al Dhafra Region of Abu Dhabi, UAE
El Tigani Ahmed El Tigani-Asil, Valeria Blanda, Ghada Elderdiri Abdelwahab, Zulaikha Mohamed Al Hammadi, Shameem Habeeba, Abdelmalik Ibrahim Khalafalla, Mohamed Ali Alhosani, Francesco La Russa, Sergio Migliore, Alessandra Torina, Guido Ruggero Loria, Sal
Animals.2021; 11(3): 666. CrossRef - Detection of Schistosoma mansoni DNA using polymerase chain reaction from serum and dried blood spot card samples of an adult population in North-western Tanzania
Antje Fuss, Humphrey D. Mazigo, Andreas Mueller
Infectious Diseases of Poverty.2021;[Epub] CrossRef - Mind the Quality Gap When Banking on Dry Blood Spots
David Carpentieri, Amber Colvard, Jackie Petersen, William Marsh, Victoria David-Dirgo, Matt Huentelman, Patrick Pirrotte, T.A. Sivakumaran
Biopreservation and Biobanking.2021; 19(2): 136. CrossRef - Long-Term Preservation and Storage of Faecal Samples in Whatman® Cards for PCR Detection and Genotyping of Giardia duodenalis and Cryptosporidium hominis
Pamela Carolina Köster, Begoña Bailo, Alejandro Dashti, Carolina Hernández-Castro, Rafael Calero-Bernal, Francisco Ponce-Gordo, David González-Barrio, David Carmena
Animals.2021; 11(5): 1369. CrossRef - Preserved Blood Spots Aid Antenatal Diagnosis of Citrullinemia Type-1
Shruti Bajaj, Uday Joglekar, Anil Jalan, Johannes Häberle, Veronique Rüfenacht
Journal of Fetal Medicine.2021; 08(02): 157. CrossRef - The genome of the zoonotic malaria parasite Plasmodium simium reveals adaptations to host switching
Tobias Mourier, Denise Anete Madureira de Alvarenga, Abhinav Kaushik, Anielle de Pina-Costa, Olga Douvropoulou, Qingtian Guan, Francisco J. Guzmán-Vega, Sarah Forrester, Filipe Vieira Santos de Abreu, Cesare Bianco Júnior, Julio Cesar de Souza Junior, Sil
BMC Biology.2021;[Epub] CrossRef - Utilization of archived neonatal dried blood spots for genome-wide genotyping
Pagna Sok, Philip J. Lupo, Melissa A. Richard, Karen R. Rabin, Erik A. Ehli, Noah A. Kallsen, Gareth E. Davies, Michael E. Scheurer, Austin L. Brown, Isabelle Chemin
PLOS ONE.2020; 15(2): e0229352. CrossRef - Ethnicity, age and disease-associated variation in body fluid-specific CpG sites in a diverse South African cohort
Farzeen Kader, Meenu Ghai, Marvellous Zhou
Forensic Science International.2020; 314: 110372. CrossRef - Gene doping and genomic science in sports: where are we?
Sheila López, João Meirelles, Vanessa Rayol, Gabriella Poralla, Nicole Woldmar, Bruna Fadel, Mariana Figueiredo, Mônica da Costa Padilha, Francisco Radler de Aquino Neto, Henrique Marcelo Gualberto Pereira, Luciana Pizzatti
Bioanalysis.2020; 12(11): 801. CrossRef - Development of novel extraction reagents for analyzing dried blood spots from crime scenes
Hae-Min Lee, Jung-Hyeon Yang, Sun-Yeong Gwon, Hee-Gyoo Kang, Sung Hee Hyun, Jiyeong Lee, Ho Joong Sung
Forensic Science International.2020; 317: 110531. CrossRef - Development of a Multiplex Loop-Mediated Isothermal Amplification (LAMP) Method for Simultaneous Detection of Spotted Fever Group Rickettsiae and Malaria Parasites by Dipstick DNA Chromatography
Lavel Chinyama Moonga, Kyoko Hayashida, Naoko Kawai, Ryo Nakao, Chihiro Sugimoto, Boniface Namangala, Junya Yamagishi
Diagnostics.2020; 10(11): 897. CrossRef - DNA Adsorption Studies of Poly(4,4′-Cychlohexylidene Bisphenol Oxalate)/Silica Nanocomposites
Aisha Nawaf Al balawi, Nor Azah Yusof, Sazlinda Kamaruzaman, Faruq Mohammad, Helmi Wasoh, Hamad A. Al-Lohedan
Materials.2019; 12(7): 1178. CrossRef - Evaluation of commercial methods to separate nucleic acids from intestinal tissues of pigs for diagnosis of porcine epidemic diarrhea
D. М. Masiuk, V. S. Nedzvetsky, A. V. Kokariev, O. V. Danchuk, T. O. Vasilenko, O. M. Yefimova
Regulatory Mechanisms in Biosystems.2019; 10(4): 477. CrossRef - Comparative analysis of three methods from dried blood spots for expeditious DNA extraction from mosquitoes; suitable for PCR based techniques
Barsa Baisalini Panda, Nitika Pradhan, Rupenangshu K. Hazra
Molecular Biology Reports.2019; 46(1): 151. CrossRef - Comparison between different methods of DNA isolation from dried blood spots for determination of malaria to determine specificity and cost effectiveness
Barsa Baisalini Panda, Arup Shankar Meher, Rupenangshu Kumar Hazra
Journal of Parasitic Diseases.2019; 43(3): 337. CrossRef - All-in-one paper-based sampling chip for targeted protein analysis
Øystein Skjærvø, Trine Grønhaug Halvorsen, Léon Reubsaet
Analytica Chimica Acta.2019; 1089: 56. CrossRef - Rapid electrophoretic recovery of DNA from dried blood spots
Mary C. Machado, Gina V. Vimbela, Michael Nilsson, Stephanie Dallaire, Rongcong Wu, Anubhav Tripathi
ELECTROPHORESIS.2019; 40(14): 1812. CrossRef - Association of adiponectin gene ( ADIPOQ ) polymorphisms with measures of obesity in Nigerian young adults
Olusegun E. Ogundele, Khalid O. Adekoya, Abraham A.A. Osinubi, Awoyemi A. Awofala, Bola O. Oboh
Egyptian Journal of Medical Human Genetics.2018; 19(2): 123. CrossRef - Biobanking in Precision Medicine
Shanavaz Nasarabadi, Michael Hogan, James Nelson
Current Pharmacology Reports.2018; 4(1): 91. CrossRef - Can malaria rapid diagnostic tests by drug sellers under field conditions classify children 5 years old or less with or without Plasmodium falciparum malaria? Comparison with nested PCR analysis
Freddy Eric Kitutu, Henry Wamani, Katarina Ekholm Selling, Fred Ashaba Katabazi, Ronald Bisaso Kuteesa, Stefan Peterson, Joan Nakayaga Kalyango, Andreas Mårtensson
Malaria Journal.2018;[Epub] CrossRef - Current Nucleic Acid Extraction Methods and Their Implications to Point-of-Care Diagnostics
Nasir Ali, Rita de Cássia Pontello Rampazzo, Alexandre Dias Tavares Costa, Marco Aurelio Krieger
BioMed Research International.2017; 2017: 1. CrossRef - Evaluating genomic DNA extraction methods from human whole blood using endpoint and real-time PCR assays
Linda Koshy, A. L. Anju, S. Harikrishnan, V. R. Kutty, V. T. Jissa, Irin Kurikesu, Parvathy Jayachandran, A. Jayakumaran Nair, A. Gangaprasad, G. M. Nair, P. R. Sudhakaran
Molecular Biology Reports.2017; 44(1): 97. CrossRef - Detection of <i>β</i>-Hemoglobin Gene and Sickle Cell Disorder from Umbilical Cord Blood
Sheikh Anika Rahman, Md. Shad Ebna Rahaman, Shahena Aktar Shipa, Md. Mohosin Rana, Rukshana Khan, Mohammad Golam Rob Mahmud, Jilwatun Noor, Firuza Sultana, Md. Faruque Miah
Journal of Biosciences and Medicines.2017; 05(10): 51. CrossRef - Dosage of Sex Chromosomal Genes in Blood Deposited on Filter Paper for Neonatal Screening of Sex Chromosome Aneuploidy
Luis Daniel Campos-Acevedo, Marisol Ibarra-Ramirez, José de Jesús Lugo-Trampe, Michelle de Jesús Zamudio-Osuna, Iris Torres-Muñoz, Ma del Roble Velasco-Campos, Luz Rojas-Patlan, Irám Pablo Rodríguez-Sánchez, Laura Elia Martínez-de-Villarreal
Genetic Testing and Molecular Biomarkers.2016; 20(12): 786. CrossRef - Single Lysis‐Salting Out Method of Genomic DNA Extraction From Dried Blood Spots
Muntaj Shaik, Devaraju Kuramkote Shivanna, Mahesh Kamate, Vedamurthy AB, Kruthika‐Vinod TP
Journal of Clinical Laboratory Analysis.2016; 30(6): 1009. CrossRef - Evaluation of artemether-lumefantrine efficacy in the treatment of uncomplicated malaria and its association with pfmdr1, pfatpase6 and K13-propeller polymorphisms in Luanda, Angola
Kinanga Kiaco, Joana Teixeira, Marta Machado, Virgílio do Rosário, Dinora Lopes
Malaria Journal.2015;[Epub] CrossRef


 PubReader
PubReader Cite
Cite
