Articles
- Page Path
- HOME > Osong Public Health Res Perspect > Volume 14(3); 2023 > Article
-
Brief Report
Temporal association between the age-specific incidence of Guillain-Barré syndrome and SARS-CoV-2 vaccination in Republic of Korea: a nationwide time-series correlation study -
Hyunju Lee1
 , Donghyok Kwon1
, Donghyok Kwon1 , Seoncheol Park2,3
, Seoncheol Park2,3 , Seung Ri Park4
, Seung Ri Park4 , Darda Chung5
, Darda Chung5 , Jongmok Ha4,6
, Jongmok Ha4,6
-
Osong Public Health and Research Perspectives 2023;14(3):224-231.
DOI: https://doi.org/10.24171/j.phrp.2023.0050
Published online: June 22, 2023
1Korea Disease Control and Prevention Agency, Cheongju, Republic of Korea
2Department of Mathematics, Hanyang University, Seoul, Republic of Korea
3Research Institute for Natural Sciences, Hanyang University, Seoul, Republic of Korea
4Gyeonggi Infectious Disease Control Center, Gyeonggi Provincial Government, Suwon, Republic of Korea
5Department of Neurology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Republic of Korea
6Department of Neurology, Yeoncheon Public Medical Center, Yeoncheon, Republic of Korea
- Corresponding author: Jongmok Ha Department of Neurology, Yeoncheon Public Medical Center, 95 Eundaeseong-ro, Jeongok-eup, Yeoncheon 11027, Republic of Korea E-mail: jongmok3245@gmail.com
© 2023 Korea Disease Control and Prevention Agency.
This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
Abstract
-
Objectives
- The incidence of Guillain-Barré syndrome (GBS) changed significantly during the coronavirus disease 2019 (COVID-19) pandemic. Emerging reports suggest that viral vector-based vaccines may be associated with an elevated risk of GBS.
-
Methods
- In this nationwide time-series correlation study, we examined the age-specific incidence of GBS from January 2011 to August 2022, as well as data on severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccinations and infections from February 2021 to August 2022. We compared the forecasted estimates of age-specific GBS incidence, using the pre-SARS-CoV-2 period as a benchmark, with the actual incidence observed during the post-vaccination period of the pandemic. Furthermore, we assessed the temporal association between GBS, SARS-CoV-2 vaccinations, and COVID-19 for different age groups.
-
Results
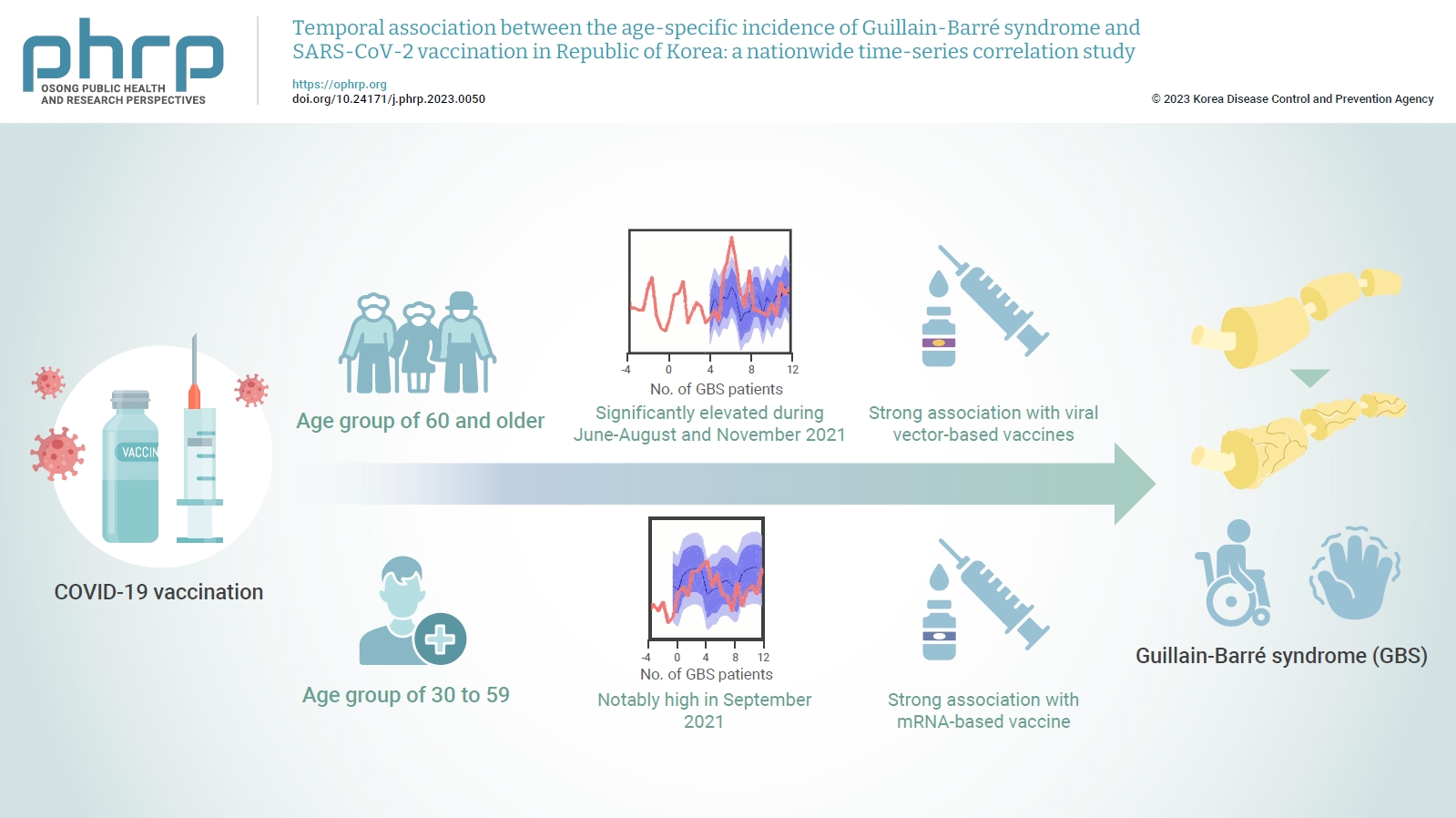
- In the age group of 60 and older, the rate ratio was significantly elevated during June-August and November 2021. A significant, strong positive association was observed between viral vector-based vaccines and GBS incidence trends in this age group (r=0.52, p=0.022). For the 30 to 59 years age group, the rate ratio was notably high in September 2021. A statistically significant, strong positive association was found between mRNA-based vaccines and GBS incidence in this age group (r=0.61, p=0.006).
-
Conclusion
- Viral vector-based SARS-CoV-2 vaccines were found to be temporally associated with an increased risk of GBS, particularly in older adults. To minimize age-specific and biological mechanism-specific adverse events, future vaccination campaigns should adopt a more personalized approach, such as recommending homologous mRNA-based SARS-CoV-2 vaccines for older adults to reduce the heightened risk of GBS.
- Since the World Health Organization declared a global pandemic in March 2020 [1], nationwide vaccination campaigns against coronavirus disease 2019 (COVID-19) have played a crucial role in achieving population-level immunity and preventing severe outcomes [2]. However, the consequences of widespread exposure to novel immune stimuli, such as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and vaccines, are still being investigated [3].
- As of August 2022, there are 3 primary vaccine mechanisms utilized in the Republic of Korea: viral vector-based ChAdOx1-S/nCoV-19 (Oxford-AstraZeneca) and Ad.26.COV2.S (Janssen); mRNA-based BNT162b2 (Pfizer-BioNTech) and mRNA-1273 (Moderna) vaccines; and the recombinant protein NVX-CoV2373 (Novavax) vaccine, which is the latest addition to the arsenal.
- In our previous study, which explored the correlation between changes in the respiratory and gastrointestinal infectious disease landscape and Guillain-Barré syndrome (GBS) incidence, we hypothesized that the significant increase in GBS from June 2021 to November 2021 might be attributed to the introduction of SARS-CoV-2 vaccines, specifically viral vector-based vaccines [4]. This hypothesis has been supported by several large database-driven studies employing prospective surveillance, retrospective cohort, or self-controlled case series study designs [5−8]. However, these methodologies do not account for seasonal patterns or incorporate historical data that could serve as a comparative baseline or reference for forecasting. Consequently, there was a need for a more intuitive approach.
- Building upon our previous research, our objectives were to (1) compare the projected age-specific GBS incidence rates with the actual incidence observed during the post-vaccination pandemic period (February 2021 to August 2022), and (2) assess the temporal relationship between GBS, SARS-CoV-2 vaccination, and COVID-19 across various age groups, as well as the biological mechanisms of the vaccines.
Introduction
- Study Design
- We carried out a nationwide longitudinal, time-series correlation study spanning from 2011 to 2022. We compared the real-world age-specific incidence of GBS during the pandemic post-vaccination period (February 2021 to August 2022) to the forecasted estimates based on the pre-vaccination period reference (January 2011 to January 2021). Furthermore, we assessed the temporal association between the incidence of GBS and either SARS-CoV-2 vaccination or COVID-19 during the post-vaccination period. The results were detailed by stratifying relevant age groups and vaccine mechanisms.
- Designation of Age Groups
- The designation of age groups was based on 2 references: vaccine rollout policy and the results from our previous study on the pandemic incidence of GBS. First, regarding vaccination policy, The Republic of Korea’s vaccine rollout during the first 2 quarters of 2021 (February to June) was primarily age-specific. The first quarter targeted patients and workers in nursing homes and facilities (age <65 years), primary medical responders in the field, and hospital personnel. The second quarter included patients and workers in nursing homes and facilities (age <65 years), residents of COVID-vulnerable facilities (homeless shelters and elder homes), and adults aged ≥60 years (initially age ≥75 years from April and age 60 to 75 years from June). Moreover, after reports of thrombosis with thrombocytopenia syndrome in younger populations, viral vector-based vaccines were contraindicated for those aged <30 years. Secondly, in relation to our previous study's results, we observed a significant increase in GBS incidence for age groups over 60. Consequently, we trichotomized the age groups as follows: 0 to 29, 30 to 59, and ≥60 years.
- Data Collection
- The National Health Insurance Service (NHIS) database was used to obtain nationwide incidence and demographic data on patients newly diagnosed with GBS, who were either hospitalized or received outpatient treatment from January 2011 to August 2022.
- In comparison to our prior research [4], we have expanded our reference timeframe from 2017–2019 to 2011–2020 to enhance the accuracy of our forecasts. Additionally, we have lengthened the vaccination periods from under one year to one and a half years to bolster our analysis.
- In patients with a primary diagnosis of GBS (International Classification of Diseases, 10th Revision: G61.0; n=12,433), there were 10,577 cases during the pre-vaccination period (January 2011 to January 2021) and 1,856 cases in the post-vaccination period (February 2021 to August 2022).
- The data on SARS-CoV-2 vaccination, such as manufacturer, dose number, and age distribution, obtained during the post-vaccination period of the pandemic, were also extracted from the NHIS database.
- The Korea Disease Control and Prevention Agency (KDCA) database was utilized to obtain nationwide SARS-CoV-2 infection data from January 2020 to August 2022 for comparison with SARS-CoV-2 vaccines. This data included all variant types and reinfection statuses.
- Statistical Analysis
- For the time-series analysis, a Gaussian regression model was chosen to predict the number of GBS patients, taking into account both the general trend and seasonality. This model was used to forecast the expected incidence and 95% prediction intervals of GBS based on the pre-vaccination reference period from January 2011 to January 2021.
- The cumulative frequency of new-onset GBS and rate ratios (RRs) between observed and expected values during the post-vaccination period of the pandemic were calculated for the total population and for specific age groups (0 to 29, 30 to 59, and ≥60 years). The RR was determined by comparing the expected value (predicted using a regression model) to the observed value (the actual number of reported GBS cases). An RR not equal to 1.0 at the 5% level was deemed clinically significant.
- We determined the temporal association between GBS and SARS-CoV-2 vaccination, stratified by biological mechanism (viral vector-based, mRNA-based, and protein recombinant vaccines), using Pearson correlation analysis. The correlation coefficient (r) was calculated to assess the trends in the absolute difference in cumulative frequency between the expected and observed cases of GBS, SARS-CoV-2 vaccination, and COVID-19. The degree of association adhered to a conventional paradigm: weak (0.10≤r<0.30), moderate (0.30≤r<0.50), and strong (r≥0.50) [9].
- Data analysis was conducted using R statistical software ver. 4.1.2 (The R Foundation). The R forecast package includes optimal values for level, trend, and seasonality parameters, which were employed in the statistical analysis of this study [10].
- Ethics Approval
- The Institutional Review Board of the KDCA exempted this study from ethical approval (2022-01-03-PE-A) because of the retrospective analysis of de-identified data that were already obtained through epidemiological investigation, presented minimal privacy or confidentiality risk to participants, and met the current public health interest requirements. All study procedures have been reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines. The requirement for consent was waived because we retrospectively collected de-identified data.
Materials and Methods
- Real-World GBS Incidence during the Pandemic Post-Vaccination Period versus Forecasted Estimates
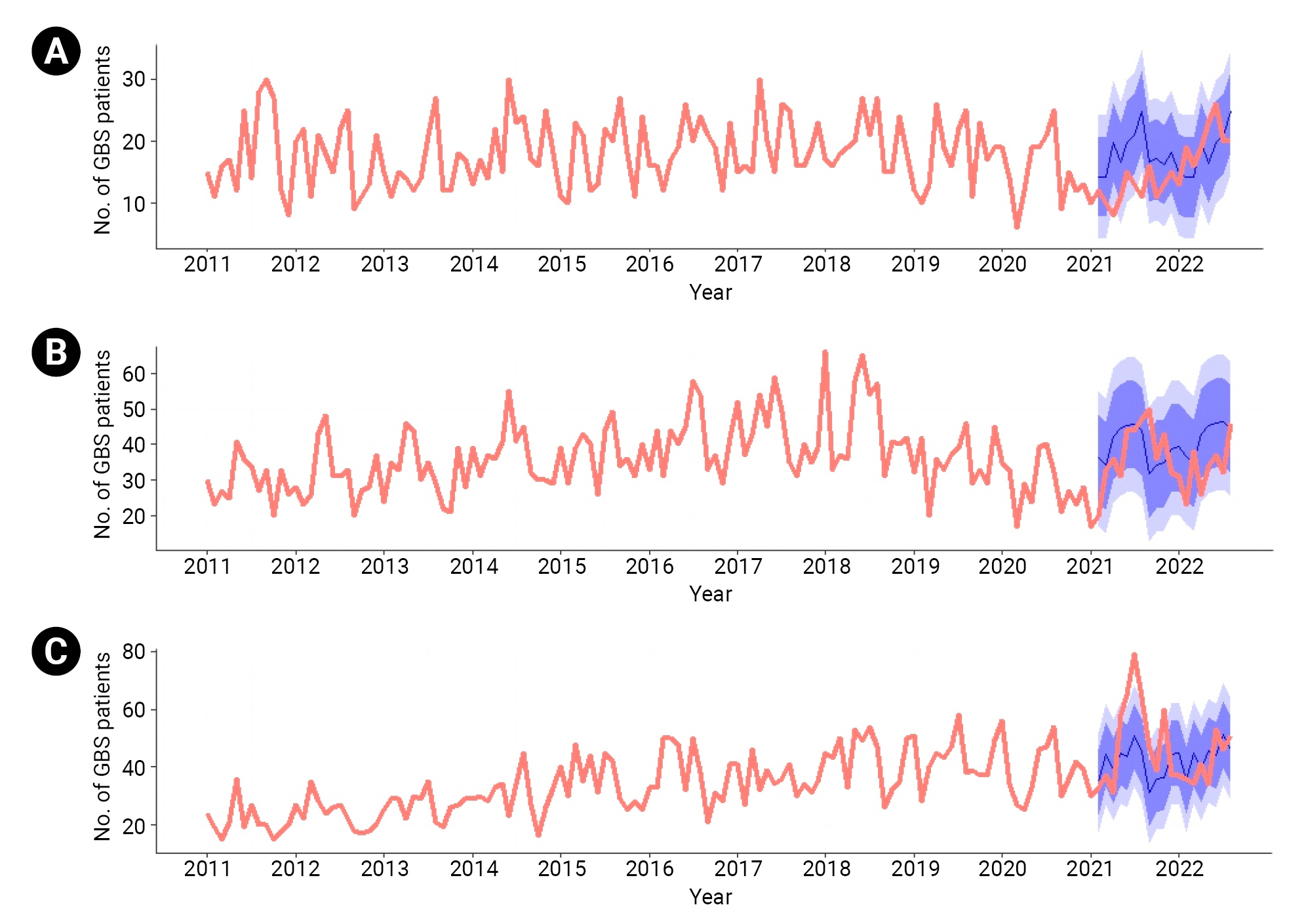
- Overall, there was no significant difference between the observed and predicted cumulative frequency of GBS for all age groups combined. However, when examined on a monthly basis, the RR was significantly elevated in September 2021 for the 30 to 59 years age group (RR, 1.57; 95% confidence interval [CI], 1.00–3.89).
- For individuals aged 60 years and older, the RR was significantly elevated in June 2021 (RR, 1.41; 95% CI, 1.08–2.03), July 2021 (RR, 1.47; 95% CI, 1.16–1.99), August 2021 (RR, 1.30; 95% CI, 1.01–1.82), and November 2021 (RR, 1.44; 95% CI, 1.07–2.19). For individuals under 30 years of age, the RR was significantly lower in April and August 2021, with RR values of 0.40 (95% CI, 0.27–0.81) and 0.44 (95% CI, 0.32–0.74), respectively. In 2022, no statistically significant differences in RR were observed across all age groups on a monthly basis (Table 1; Figure 1).
- Time-Series Correlation Analysis during the Pandemic Post-Vaccination Period
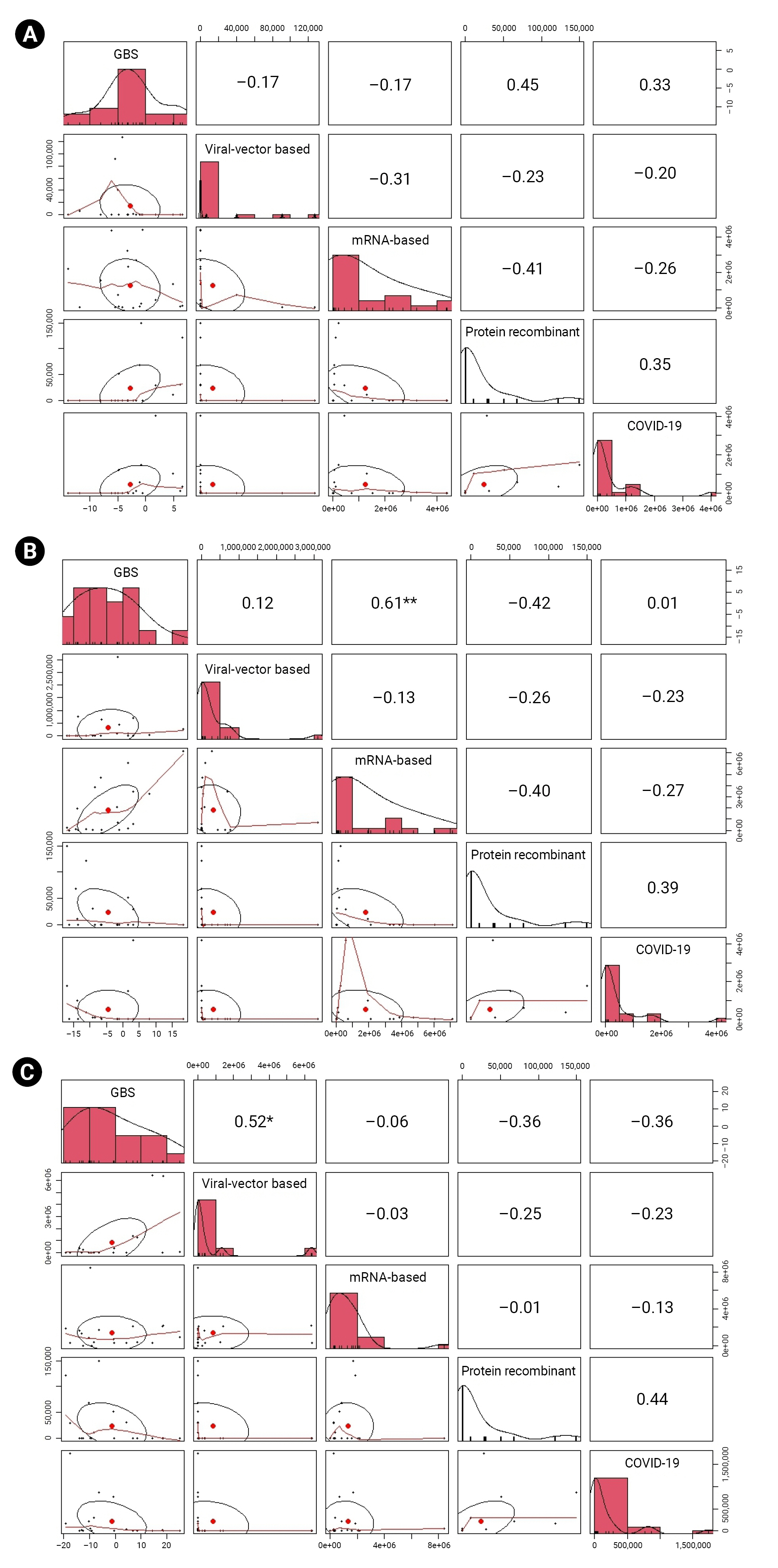
- We performed Pearson correlation analysis to analyze the time-series relationships of GBS with SARS-CoV-2 vaccination and COVID-19 incidence during the post-vaccination phase of the pandemic (February 2021 to August 2022).
- For individuals under 30 years of age, no significant temporal association was found between GBS and either SARS-CoV-2 vaccination or COVID-19 infection. In the 30 to 59 age group, a statistically significant and strong positive temporal association was observed between the incidence of GBS and mRNA-based vaccines (r=0.61, p=0.006). No significant associations were found for other vaccine mechanisms or COVID-19 infections. In older adults (≥60 years), viral vector-based vaccines demonstrated a significant and strong positive temporal association with the incidence of GBS (r=0.52, p=0.022). However, no significant temporal association was observed between GBS incidence and mRNA-based vaccines (r=–0.06, p=0.797), recombinant protein vaccines (r=–0.36, p=0.126), or COVID-19 infection (r=–0.36; p=0.127) (Figure 2).
Results
- We conducted a nationwide time-series correlation study involving 1,856 cases of GBS during the pandemic following the vaccine rollout. We compared the real-world age-specific incidence of GBS with forecasted values based on pre-pandemic incidence data. Additionally, we assessed the temporal association between the time-series trends of GBS and SARS-CoV-2 vaccination, stratified into 3 categories of biological mechanisms. Our main findings include: (1) a significantly high number of GBS patients among older adults in June to August and November 2021 compared to forecasted estimates; (2) the observed incidence of GBS converging with the forecasted incidence in 2022 for all age groups; and (3) a relevant significant temporal association between viral vector-based vaccine and GBS incidence in older adults. We believe that our study supports the findings from large database-driven studies and demonstrates the utility of time-series analysis as an auxiliary tool in evaluating temporal associations. This follow-up study also validates and expands upon the speculations made regarding the temporal association between vaccination and GBS in our previous study.
- Regarding vaccination rates among different age groups, individuals under 30 years of age primarily received mRNA-based vaccines (98.7%) during the post-vaccination period of the pandemic. In the 30 to 59 age group, the majority of participants were also vaccinated with mRNA-based vaccines (89.6%), while a smaller percentage received viral vector-based vaccines (10.1%). For those aged 60 and older, mRNA-based vaccines remained the predominant choice (59.4%); however, this group had the highest proportion of individuals receiving viral-vector vaccines (39.6%) (Table S1).
- Our results reveal varying findings in GBS incidence among different age groups, with a particular focus on older adults. Although age is a recognized risk factor for GBS [11,12], our estimates indicate that even after accounting for age and comparing it to the projected 2021 incidence based on historical baselines, this result remained valid. Furthermore, despite the influx of new vaccine recipients in 2022, GBS incidence aligned with the predicted outcomes. This implies that the 2021 increase in GBS incidence among older adults cannot be solely attributed to age, but was also influenced by other factors closely related to age. Considering the results of the mechanism-stratified time-series correlation analyses in conjunction with this finding, we can infer that viral vector-based vaccines may have a temporal association with increased GBS incidence. This observation supports previous studies that suggested a potential link between viral vector-based vaccines [5,6]. Additionally, while COVID-19 remains a relevant factor during the pandemic era that has a temporal association with GBS [13], the results of the correlation analysis were statistically insignificant, which is consistent with the findings from our previous study.
- In September 2021, there was a notable increase in the incidence of GBS among individuals aged 30 to 59 years compared to predicted values. Moreover, a strong positive correlation was observed between mRNA-based vaccines and GBS incidence in this age group. Although this finding has not been replicated in other studies, we hypothesize that it may be due to the impact of heterologous vaccination in the 30 to 59 age group, which elicits greater immunogenicity and reactogenicity compared to homologous vaccination methods [14]. In Korea, most recipients of heterologous vaccines were initially vaccinated with viral vector-based vaccines and later received mRNA-based vaccines. Consequently, individuals aged 30 to 59 who were vaccinated with viral vector-based vaccines (primarily ChAdOx1-S/nCoV-19) in June were subsequently given mRNA-based vaccines (predominantly BNT162b2) from August to September 2021 [15]. In contrast, the vaccination rate for those aged 0 to 29 was lower than in other age groups, and the majority received mRNA-based vaccines, resulting in an insignificant temporal association between GBS and vaccination in this younger population. Overall, we hypothesize that the age-stratified incidence of GBS reflects a dose-response relationship with vector-based vaccines. However, additional research is needed to confirm this relationship and better understand the underlying mechanism.
- Our study has several notable limitations: first, as an observational study, it does not establish causation. However, a potential dose-response relationship and the reproducibility of findings from different study designs may support causality. Nonetheless, GBS remains a rare disease, and the risk-to-benefit ratio should always be considered. Second, our study collected data on a per-dose basis, without gathering individual vaccination information. Consequently, we did not account for homologous or heterologous vaccination, which may have varying immunogenicity and reactogenicity [16,17]. Future studies addressing the cumulative effects of immune exposure should be conducted to address this issue. Third, we did not consider changes in vaccination policies throughout the study period. This may have led to minor fluctuations in vaccination patterns across age groups, but the overall impact is unclear. Relatedly, our age group structure may not accurately reflect vaccination policies, as our age groups were reconstituted from the 10-year interval data used in our previous study. To improve accuracy, future studies employing 5-year age gaps may be beneficial. Finally, our study focuses on a single ethnicity. However, the Asian population has been underrepresented in phase III trials of SARS-CoV-2 vaccines, making it crucial to collect specific adverse event data from this demographic.
- In conclusion, we hypothesize that a synergistic effect between age and the viral vector-based SARS-CoV-2 vaccine mechanism may contribute to an increased risk of GBS. We propose that future vaccination campaigns adopt a more personalized approach, or “safest pathway,” to minimize age-specific and biological mechanism-specific adverse events. For example, recommending mRNA-based vaccines instead of viral vector-based platforms and performing homologous vaccination whenever possible in older adults aged 60 years and above could be considered, until proven otherwise by other population-based studies, to avoid an increased risk of post-vaccination GBS.
Discussion
- • The real-world incidence of Guillain-Barré syndrome (GBS) in the Republic of Korea was significantly higher than the forecasted estimates in June to August and November 2021 in older adults, while in 2022, the incidence remained within the estimated range regardless of age.
- • Viral vector-based vaccines were temporally associated with GBS incidence in older adults. A dose-response relationship may exist between the two.
- • A synergistic effect between age and the vaccine mechanism may have contributed to the increased risk of GBS, warranting a more personalized approach to mass vaccination campaigns.
HIGHLIGHTS
Supplementary Material
Table S1.
-
Ethics Approval
The Institutional Review Board of the KDCA exempted this study from ethical approval (2022-01-03-PE-A) because of the retrospective analysis of de-identified data that were already obtained through epidemiological investigation, presented minimal privacy or confidentiality risk to participants, and met the current public health interest requirements. The requirement for consent was waived because we retrospectively collected de-identified data.
-
Conflicts of Interest
The authors have no conflicts of interest to declare.
-
Funding
None.
-
Availability of Data
The datasets are not publicly available. Any inquiries regarding the study should be directed to the corresponding authors.
-
Authors’ Contributions
Conceptualization: HL, SRP, DC; Data curation: HL, DK; Formal analysis: HL, SP; Methodology: HL, SRP, JH; Validation: DK; Visualization: HL, SP; Writing–original draft: HL, JH; Writing–review & editing: DC, JH. All authors read and approved the final manuscript.
-
Acknowledgements
The authors acknowledge the assistance of all their colleagues at the Korea Disease Control and Prevention Agency and Gyeonggi Infectious Disease Control Center for their dedication to public health and overall support for this project.
Article information


| Date |
Age <30 y |
Age 30–59 y |
Age ≥60 y |
||||||
|---|---|---|---|---|---|---|---|---|---|
| Obs. | Exp. | RR (95% CI)a) | Obs. | Exp. | RR (95% CI) | Obs. | Exp. | RR (95% CI) | |
| Total | 291 | 344 | 0.85 (0.54–1.89) | 683 | 769 | 0.89 (0.61–1.67) | 882 | 907 | 0.97 (0.75–1.38) |
| 2021 | 135 | 199 | 0.68 (0.44–1.51) | 416 | 432 | 0.96 (0.65–1.86) | 550 | 503 | 1.09 (0.83–1.58) |
| February | 12 | 14 | 0.84 (0.50–2.83) | 20 | 36 | 0.55 (0.36–1.16) | 33 | 42 | 0.78 (0.59–1.18) |
| March | 10 | 14 | 0.70 (0.41–2.36) | 33 | 34 | 0.96 (0.62–2.16) | 37 | 49 | 0.75 (0.58–1.05) |
| April | 8 | 20 | 0.40 (0.27–0.81)* | 36 | 42 | 0.85 (0.59–1.53) | 31 | 45 | 0.69 (0.52–1.01) |
| May | 11 | 17 | 0.67 (0.42–1.68) | 31 | 45 | 0.70 (0.49–1.21) | 58 | 51 | 1.14 (0.89–1.57) |
| June | 15 | 20 | 0.75 (0.50–1.51) | 44 | 46 | 0.97 (0.68–1.66) | 65 | 46 | 1.41 (1.08–2.03)* |
| July | 13 | 21 | 0.62 (0.42–1.17) | 44 | 46 | 0.96 (0.68–1.64) | 79 | 54 | 1.47 (1.16–1.99)* |
| August | 11 | 25 | 0.44 (0.32–0.74)* | 47 | 44 | 1.07 (0.75–1.88) | 64 | 49 | 1.30 (1.01–1.82)* |
| September | 16 | 17 | 0.96 (0.60–2.41) | 50 | 32 | 1.57 (1.00–3.89)* | 47 | 39 | 1.22 (0.89–1.93) |
| October | 11 | 17 | 0.64 (0.41–1.54) | 36 | 34 | 1.05 (0.68–2.33) | 39 | 40 | 0.99 (0.73–1.54) |
| November | 13 | 16 | 0.80 (0.50–2.08) | 43 | 35 | 1.23 (0.80–2.69) | 60 | 42 | 1.44 (1.07–2.19)* |
| December | 15 | 18 | 0.82 (0.53–1.80) | 32 | 39 | 0.82 (0.55–1.61) | 37 | 47 | 0.79 (0.61–1.14) |
| 2022 | 156 | 145 | 1.07 (0.69–2.39) | 267 | 336 | 0.79 (0.55–1.45) | 332 | 403 | 0.82 (0.64–1.15) |
| January | 13 | 15 | 0.88 (0.53–2.73) | 31 | 39 | 0.79 (0.53–1.51) | 37 | 50 | 0.74 (0.57–1.03) |
| February | 19 | 14 | 1.34 (0.78–4.56) | 23 | 37 | 0.63 (0.41–1.30) | 36 | 44 | 0.81 (0.61–1.19) |
| March | 16 | 14 | 1.13 (0.66–3.84) | 38 | 35 | 1.09 (0.71–2.41) | 34 | 52 | 0.66 (0.51–0.91) |
| April | 19 | 20 | 0.96 (0.64–1.95) | 26 | 43 | 0.60 (0.42–1.09) | 41 | 47 | 0.87 (0.67–1.24) |
| May | 23 | 17 | 1.39 (0.87–3.56) | 34 | 45 | 0.75 (0.53–1.30) | 34 | 53 | 0.64 (0.50–0.87) |
| June | 26 | 20 | 1.31 (0.87–2.63) | 37 | 46 | 0.80 (0.57–1.37) | 53 | 49 | 1.09 (0.84–1.55) |
| July | 20 | 21 | 0.95 (0.64–1.81) | 32 | 46 | 0.69 (0.49–1.17) | 46 | 56 | 0.82 (0.65–1.10) |
| August | 20 | 25 | 0.80 (0.57–1.35) | 46 | 45 | 1.03 (0.72–1.81) | 51 | 52 | 0.99 (0.77–1.36) |
- 1. Liu Q, Xu K, Wang X, et al. From SARS to COVID-19: what lessons have we learned? J Infect Public Health 2020;13:1611−8.ArticlePubMedPMC
- 2. Yi S, Choe YJ, Lim DS, et al. Impact of national COVID-19 vaccination Campaign, South Korea. Vaccine 2022;40:3670−5.ArticlePubMedPMC
- 3. Hwang I, Park K, Kim TE, et al. COVID-19 vaccine safety monitoring in Republic of Korea from February 26, 2021 to October 31, 2021. Osong Public Health Res Perspect 2021;12:396−402.ArticlePubMedPMCPDF
- 4. Lee H, Heo N, Kwon D, et al. Deciphering changes in the incidence of the Guillain-Barre syndrome during the COVID-19 pandemic: a nationwide time-series correlation study. BMJ Neurol Open 2022;4:e000378.ArticlePubMedPMC
- 5. Patone M, Handunnetthi L, Saatci D, et al. Neurological complications after first dose of COVID-19 vaccines and SARS-CoV-2 infection. Nat Med 2021;27:2144−53.ArticlePubMedPMCPDF
- 6. Hanson KE, Goddard K, Lewis N, et al. Incidence of Guillain-Barre syndrome after COVID-19 vaccination in the Vaccine Safety Datalink. JAMA Netw Open 2022;5:e228879.ArticlePubMedPMC
- 7. Garcia-Grimshaw M, Galnares-Olalde JA, Bello-Chavolla OY, et al. Incidence of Guillain-Barre syndrome following SARS-CoV-2 immunization: analysis of a nationwide registry of recipients of 81 million doses of seven vaccines. Eur J Neurol 2022;29:3368−79.ArticlePubMed
- 8. Wong CK, Lau KT, Xiong X, et al. Adverse events of special interest and mortality following vaccination with mRNA (BNT162b2) and inactivated (CoronaVac) SARS-CoV-2 vaccines in Hong Kong: a retrospective study. PLoS Med 2022;19:e1004018.ArticlePubMedPMC
- 9. Parker RI, Hagan-Burke S. Useful effect size interpretations for single case research. Behav Ther 2007;38:95−105.ArticlePubMed
- 10. Hyndman RJ, Khandakar Y. Automatic time series forecasting: the forecast package for R. J Stat Softw 2008;27:1−22.Article
- 11. Bragazzi NL, Kolahi AA, Nejadghaderi SA, et al. Global, regional, and national burden of Guillain-Barre syndrome and its underlying causes from 1990 to 2019. J Neuroinflammation 2021;18:264. ArticlePubMedPMC
- 12. Kim AY, Lee H, Lee YM, et al. Epidemiological features and economic burden of Guillain-Barre syndrome in South Korea: a nationwide population-based study. J Clin Neurol 2021;17:257−64.ArticlePubMedPMCPDF
- 13. Palaiodimou L, Stefanou MI, Katsanos AH, et al. Prevalence, clinical characteristics and outcomes of Guillain-Barre syndrome spectrum associated with COVID-19: a systematic review and meta-analysis. Eur J Neurol 2021;28:3517−29.ArticlePubMedPMCPDF
- 14. Klemis V, Schmidt T, Schub D, et al. Comparative immunogenicity and reactogenicity of heterologous ChAdOx1-nCoV-19-priming and BNT162b2 or mRNA-1273-boosting with homologous COVID-19 vaccine regimens. Nat Commun 2022;13:4710. ArticlePubMedPMCPDF
- 15. Korea Disease Control and Prevention Agency (KDCA). February 14, 2023 COVID-19 vaccination [Internet]. KDCA; 2023 [cited 2023 Feb 19]. Available from: https://ncov.kdca.go.kr/tcmBoardList.do?brdId=&brdGubun=&dataGubun=&ncvContSeq=&contSeq=&board_id=140&gubun=. Korean.
- 16. Barros-Martins J, Hammerschmidt SI, Cossmann A, et al. Immune responses against SARS-CoV-2 variants after heterologous and homologous ChAdOx1 nCoV-19/BNT162b2 vaccination. Nat Med 2021;27:1525−9.ArticlePubMedPMCPDF
- 17. Khoo NK, Lim JM, Gill US, et al. Differential immunogenicity of homologous versus heterologous boost in Ad26.COV2.S vaccine recipients. Med 2022;3:104−18.ArticlePubMedPMC
References
Figure & Data
References
Citations

- mRNA-LNP COVID-19 Vaccine Lipids Induce Complement Activation and Production of Proinflammatory Cytokines: Mechanisms, Effects of Complement Inhibitors, and Relevance to Adverse Reactions
Tamás Bakos, Tamás Mészáros, Gergely Tibor Kozma, Petra Berényi, Réka Facskó, Henriette Farkas, László Dézsi, Carlo Heirman, Stefaan de Koker, Raymond Schiffelers, Kathryn Anne Glatter, Tamás Radovits, Gábor Szénási, János Szebeni
International Journal of Molecular Sciences.2024; 25(7): 3595. CrossRef - Guillain–Barre syndrome following COVID-19 vaccination: a study of 70 case reports
Biki Kumar Sah, Zahra Fatima, Rajan Kumar Sah, Bushra Syed, Tulika Garg, Selia Chowdhury, Bikona Ghosh, Binita Kunwar, Anagha Shree, Vivek Kumar Sah, Anisha Raut
Annals of Medicine & Surgery.2024; 86(4): 2067. CrossRef


 Cite
Cite