Articles
- Page Path
- HOME > Osong Public Health Res Perspect > Volume 11(5); 2020 > Article
-
Original Article
Alarm Thresholds for Pertussis Outbreaks in Iran: National Data Analysis - Yousef Alimohamadia, Seyed Mohsen Zahraeib, Manoochehr Karamic, Mehdi Yaseria, Mojtaba Lotfizadd, Kourosh Holakouie-Naienia
-
Osong Public Health and Research Perspectives 2020;11(5):309-318.
DOI: https://doi.org/10.24171/j.phrp.2020.11.5.07
Published online: October 22, 2020
aDepartment of Epidemiology and Biostatistics, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran
bCenter for Communicable Diseases Control, Ministry of Health and Medical Education, Tehran, Iran
cResearch Center for Health Sciences, Hamadan University of Medical Sciences, Hamadan, Iran
dSchool of Electrical and Computer Engineering, Tarbiat Modares University, Tehran, Iran
- *Corresponding author: Kourosh Holakouie-Naieni, Department of Epidemiology and Biostatistics, School of Public Health, Tehran University of Medical Sciences, Ghods Street, Enghelab Square, P.O. Box 1417613151, Tehran, Iran, E-mail: holakoik@hotmail.com
• Received: April 11, 2020 • Revised: July 14, 2020 • Accepted: July 16, 2020
Copyright ©2020, Korea Centers for Disease Control and Prevention
This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
Abstract
-
Objectives
- The purpose of the current study was to determine the upper threshold number of cases for which pertussis infection would reach an outbreak level nationally in Iran.
-
Methods
- Data on suspected cases of pertussis from the 25th February 2012 to the 23rd March 2018 from the Center for Disease Control and Prevention in Iran was used. The national upper threshold level was estimated using the exponentially weighted moving average (EWMA) method and the Poisson regression method.
-
Results
- In total, 2,577 (33.6%) and 1,714 (22.3%) cases were reported in the Spring and Summer respectively. There were 1,417 (18.5%) and 1,971 (25.6%) cases reported in the Autumn and Winter, respectively. The overall upper threshold using the EWMA and the Poisson regression methods, was estimated as a daily occurrence of 8 (7.55) and 7.50 (4.48–11.06) suspected cases per 10,000,000 people, respectively. The daily seasonal thresholds estimated by the EWMA and the Poisson regression methods were 10, 7, 6, 8 cases and 10, 7, 7, 9 cases for the Spring, Summer, Autumn, and Winter, respectively.
-
Conclusion
- The overall and seasonal estimated thresholds by the 2 methods were similar. Therefore, the estimated thresholds of 6–10 cases in a day, per 10,000,000 people could be used to detect pertussis outbreaks and epidemics by health policymakers.
- Pertussis is a notifiable infectious disease caused by Bordatella pertussis which is characterized by a paroxysmal cough [1,2]. Despite global vaccination against pertussis, the reported number of cases are increasing, and it can be considered as a reemerging infectious disease [3,4]. Although pertussis can occur in all age groups, infants less than 6 months have the highest morbidity due to infection [5–7]. Clinical symptoms of pertussis are highly dependent upon the age of the patient and the resilience of their immune system [8,9]. The severity of symptoms, hospitalization, and mortality are more notable among infants [10]. The intensity of disease severity is often mild in adults however, this group is the main source of transmission of infection to infants and neonates [11,12]. According to the global burden of disease study in 2013, pertussis-related death accounted for nearly 400 deaths per million births during the first year of life [13]. In 2014, a global estimation showed that there were nearly 24.1 million pertussis cases and 160,700 deaths related to pertussis which occurred in children under 5 years [14]. Since the mid-twentieth century, global efforts to control/eradicate pertussis began by implementing widespread immunization programs [15,16]. Before immunization programs were in place, the typical time interval between outbreaks of pertussis was every 2 to 3 years which increased to 3 to 4 years following the implementation of comprehensive vaccination programs [11,17].
- Because of the weakening of vaccine immunity over time, the immunity induced by the pertussis vaccine is temporary [18], hence, infection in the susceptible population, especially the young, will always increase. Subsequently a drop in the level of herd immunity concurrent with migration of people from other countries could be the cause of outbreaks of pertussis. In spite of the increasing trend of registered suspected cases of pertussis in Iran [19], there is no information on the level of cases in an outbreak which would classify the outbreaks as a national epidemic threshold.
- There are many methods to determine the threshold number of cases of infectious diseases to cause alarm including absolute values/standard thresholds, relative increase, statistical cut-off points, and upper control limits of outbreak detection methods [20]. The Poisson regression model is an appropriate model for a rare response without normal distribution [21]. The exponentially weighted moving average (EWMA) method is a model used for individual observations. This algorithm is weighted average of all past and new data. The Poisson regression and the EWMA methods of estimation are accepted models in the field of infection outbreak detection and define the threshold level where there is cause for alarm. These methods are easy to implement and were used to determine the upper threshold number of cases or the “alarm threshold” for pertussis infection in Iran per 10,000,000 people.
Introduction
- 1. Data source
- The data was retrieved from the Department of Vaccine-Preventable Diseases at the Ministry of Health, Iran. This national data included all daily suspected cases of pertussis from the 25th February 2012 to the 23rd March 2018. Pertussis surveillance in Iran is based upon suspected cases.
- A suspected case of pertussis was defined as a patient with clinical signs of persistent coughing lasting for 2 weeks and 1 of the following classical symptoms: (a) paroxysmal coughing, (b) inspiratory whooping, or (c) posttussive vomiting. In pertussis surveillance, nasopharyngeal swabs are taken from all suspected cases of pertussis and transported to the Iranian national reference laboratory within 72 hours [in Bordetella-specific (Bordet-Gengou) transport media] to be confirmed by laboratory-based diagnostic methods. The isolation of pertussis in samples was performed using PCR. The sensitivity and specificity of PCR detection of pertussis has previously been estimated to be approximately 100% and 97% respectively [22,23].
- A confirmed case of pertussis was defined as a case with clinical symptoms whose diagnosis had been confirmed by a laboratory [11]. All suspected clinical cases that have not been tested are classified as cases. All confirmed cases are treated. Prophylaxis treatment is recommended for contact cases.
- 2. Methods to establish upper control limits or the “alarm thresholds”
- The statistic of the EWMA at time t was defined as follows:
- Where Yt equals the number of suspected cases of pertussis in a day of t and λ is the smoothing parameter which includes 0 < λ ≤ 1.
- By using this method, the upper control limit or the “alarm threshold” level was calculated as follows:
- Where k is specified in a way that results in the desired confidence interval. In the current study, the k = 2 or 95% confidence interval was considered as the desired confidence interval [20,24]. σEWMA was the standard deviation of the calculated statistics of the EWMA at times t to tn and EWMA0 was the mean of reported cases in non-outbreak days. In other words, EWMA0 and σEWMA were the means and standard deviations on the baseline or historical data. In this method when the EWMA statistic was more than the upper control limit, it was considered as an “alarm” or aberration.
- Due to the lack of known outbreak periods in the dataset, the non-outbreak period was defined. Firstly, the mean (μ) and standard deviation (δ) of understudy data were calculated. The period with an amount less than μ+2δ were considered as a non-outbreak period or target period.
- To determine the upper control limit, in the first step, the expected mean of suspected pertussis cases per day was estimated as follows:
- Where μt was the expected mean of occurred cases at time t, and β was the coefficient of X, and X is another factor, which are effective factors on the expected mean of pertussis cases. In other words, μt is the dependent variable and X is the independent or predictor variable. In the current study the expected mean was determined after adjusting for day and week effects, by entering these factors in the model, and the seasonal effect was adjusted by estimating seasonal thresholds. After the estimation of expected mean of occurred cases at time t (μt), the “alarm threshold” limit was estimated as follow:
- Where Vt was the variance of estimated mean and it was equal to:
- According to the “alarm threshold” limit formula (Formula 4), the estimation of the “alarm threshold” level needed 2 parameters that included μt (expected number or mean of occurred cases at time t) and Vt (variance of the estimated mean) which were calculated according to Formula 3 and Formula 5, respectively. In Formula 3 the dependent or response variable was the mean of expected cases at time t which was calculated from the observed cases. After the estimation of μt and Vt, the level of “alarm threshold” for time t was calculated according to Formula 4. However, the dataset used was time-series including a series of count data over time (for example 3, 4, 8, 9, 11, 12 cases for different days). In this model, by considering the reported cases (reported counts) in time t1 to tn the expected mean (count) or “alarm thresholds” for the mentioned time interval, were calculated.
- After the calculation of the “alarm threshold” limit for each day, because of entering the day and week effect into the model, there was a wide range of estimated thresholds in different days, so the median of calculated limits was considered as an overall threshold. Moreover, the 2.5 and 97.5 percentiles of the calculated limits were considered as 95% coverage for the estimated “alarm threshold.” If the number of reported cases was more than upper control limit it can be considered as an “alarm” or aberrations.
- The defined level of “alarm threshold” was affected by the incidence of reported cases. Due to heterogeneity in incidence of suspected pertussis cases in different provinces of Iran, it was decided that a definition of strata (each stratum includes provinces with similar incidence) was needed and the “alarm threshold” was calculated in the different stratums. Using SPSS, the average incidence of suspected cases per 100,000 for all provinces within the study period was classified into 3 strata that included high (> 3 cases), moderate (1–3 cases) and low (< 1 case) per 100,000. Cluster 1 included Zanjan, Qom, Qazvin, Mazandaran. Cluster 2 included Yazd, Semnan, Kurdistan, Khuzestan, South-Khorasan, North-Khorasan, Kermanshah, Ilam, Hamadan, Golestan, Isfahan, East-Azerbaijan, Chaharmahal, Busher, Alborz, and Tehran. Cluster 3 included West-Azerbaijan, Sistan Baluchestan, Markazi, Lorestan, Kohgiluyeh, Razavi-Khorasan, Kerman, Hormozgan, Gilan, Fars, and Ardabil.
- In Iran, the 21st March is the first day of the year and Spring begins on the 21st March until the 21st June. Summer begins on the 22nd June and lasts until the 22nd September. Autumn is from the 23rd September until the 21st December and Winter begins on the 22nd December and ends on the 20th March.
- To adjust for the seasonal effect, the seasonal specific thresholds were defined. Due to different population sizes in each stratum, the defined “alarm threshold” can be affected by population size in each stratum, so to adjust for this effect, the calculated “alarm threshold” in each stratum was divided into its population size and multiplied by 10,000,000 people. Because of the low incidence of pertussis in Iran to obtain tangible quantities for the threshold, the calculation in each stratum was performed per 10 million people in the national population.
- All the statistical analyses to determine the “alarm threshold” were performed using Microsoft Excel 2010. To compute the expected mean using the Poisson regression method, Stata software Version 15 (StataCorp, College Station, TX, USA) was used.
Materials and Methods
2.1. Upper control limits of the EWMA method
2.2. Definition of the non-outbreak period
2.3. Upper control limit of the Poisson regression method
2.4. Stratum definition
2.5. Season period
2.6. Adjusting for seasonal effect and population size
2.7. Statistical analysis
- 1. Descriptive statistics
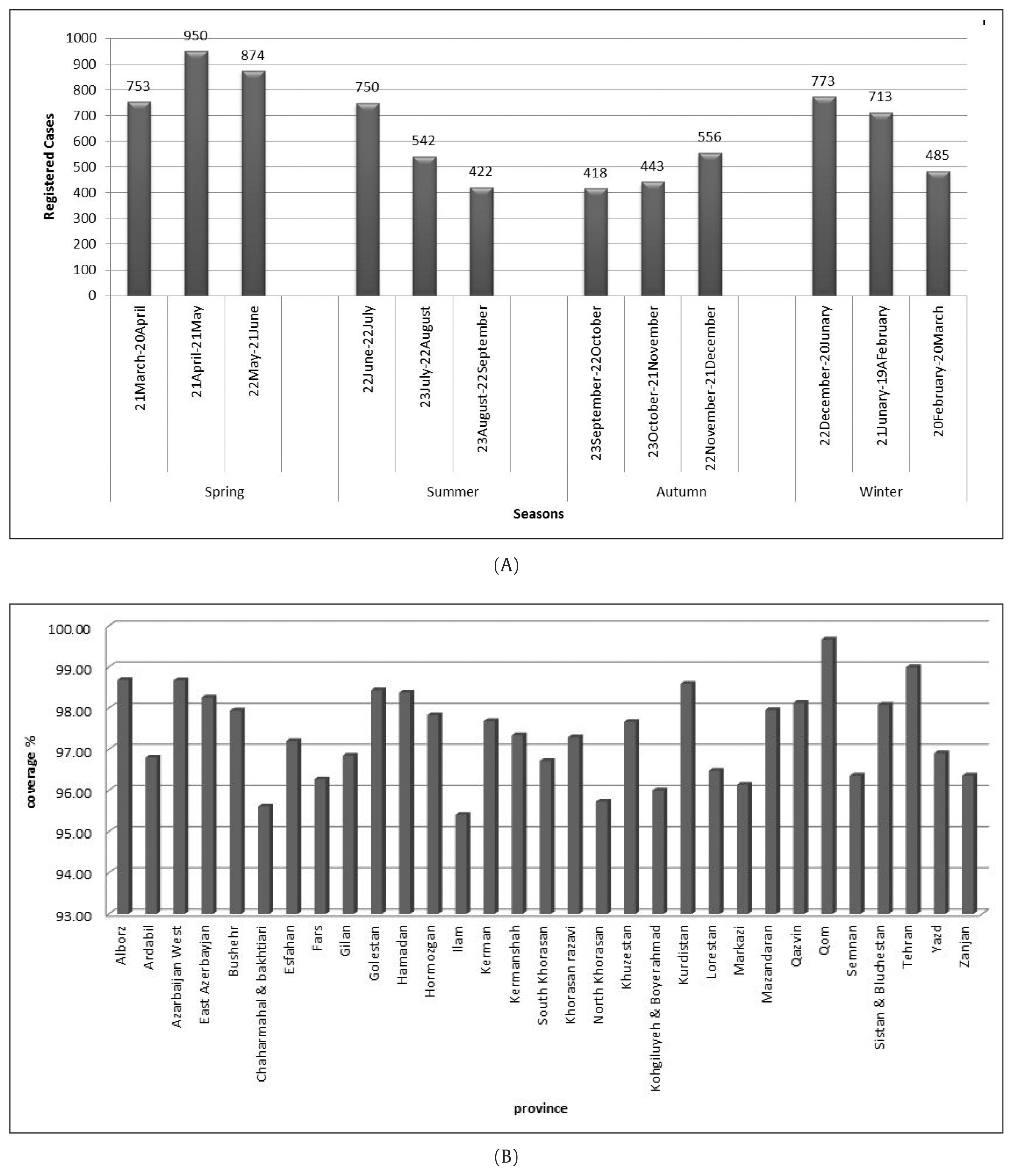
- From the 25th February 2012 to the 23rd March 2018, there were 7,679 total cases of pertussis with an exact date of reporting registered across the country, with 7,553 (98.3%) having Iranian nationality. There were 4,838 (63%) of total cases < 1 year and 2,841 (37%) of total cases were > 1 year. Overall, there were 2,577 (33.6%) cases reported in Spring, 1,714 (22.3%) cases reported in Summer, 1,417 (18.5%) cases reported in Autumn and 1,971 (25.6%) cases reported in Winter. The highest number of suspected cases of pertussis was reported during the period of the 21st April to the 21st June and the lowest number of cases was reported from the 21st September to the 20th October (Figure 1A). The mean (standard deviation) number of daily reported cases was 3.46 (2.86) from 2012 to 2018 at the national level (range = 0–23). The median, mode, and 3rd quartile of the overall number of suspected and confirmed cases of pertussis regardless of the age of cases were 3, 2, and 5, respectively. Other descriptive statistics according to the season and age of cases are shown in Table 1. Moreover, the coverage of the third pentavalent vaccine, also known as Pent3 (including diphtheria, tetanus, pertussis, hepatitis B and Hemophilus Influenza type-b) was more than 95% in the Iranian population on 2017. More information is provided in Figure 1B.
- 2. The EWMA threshold for aberration detection
- The “alarm threshold” for the EWMA was defined as a daily occurrence of 8 (7.55) suspected pertussis cases. In other words, if the number of reported cases in a day exceeded 8, it must be considered as an outbreak or aberration or “alarm.” Considering the alarming level of 7.55, there were about 190 days aberrations from the “alarm threshold” for the reported data (Figure 2A). According to this method, the “alarm thresholds” for Spring, Summer, Autumn, and Winter were estimated as 10, 7, 6 and 8 of suspected cases, respectively. The level determined to be the “alarm threshold” according to different age groups is shown in Table 2. The estimated “alarm threshold” according to population size is shown in Table 3. The “alarm thresholds” in the first, second, and third stratum were estimated as 3, 5, and 2 cases per day, respectively. The “alarm threshold” in the second stratum was higher than the first stratum due to a larger population size in the former. However, after adjustment for the population size by calculating the “alarm threshold” per 10,000,000 people, the “alarm threshold” in the first stratum with a higher incidence of suspected cases, was higher than the other strata. These “alarm thresholds” for the first, second, and third stratum were 4, 2, and 1 per 10,000,000, respectively (Table 3).
- 3. The Poisson regression model
- The “alarm threshold” for the Poisson regression was defined as the daily occurrence of 7.50 (4.48–11.06) suspected pertussis cases. In other words, if the number of reported cases in a day exceeded 8 cases (95% coverage: 4.48–11.06), it must be considered as an outbreak. Considering the estimated range for the “alarm threshold,” there were about 140 days’ aberrations from the “alarm threshold” (Figure 2B). Accordingly, the level for the “alarm threshold” for the Spring, Summer, Autumn, and Winter was estimated as 10, 7, 7, and 9 cases of suspected pertussis, respectively. The determined level of “alarm threshold” according to different age groups is shown in Table 2. The “alarm thresholds” in the first, second, and third stratum were 4, 6, and 2 cases per day. The “alarm threshold” in the second stratum was higher than the first stratum due to the larger population size in the former. Nevertheless, after adjustment on the “alarm threshold” per 10,000,000 people, the “alarm threshold” in the first stratum with a higher incidence of suspected cases was higher than the other strata. The “alarm thresholds” for the first, second, and third stratum were 6, 2, and 1 per 10,000,000, respectively (Table 3).
Results
- Identification and management of pertussis outbreaks can be very difficult because of similar symptoms between pertussis and other respiratory infections. There is a lack of an upper threshold for pertussis outbreaks which would raise an “alarm threshold” to the health authorities in Iran.
- There are many methods for “alarm thresholds” for the detection of outbreaks [26–29]. In the current study, the EWMA and the Poisson regression models were used. The EWMA algorithm has been used to determine and define the “alarm threshold” for outbreak detection of other infectious diseases in Iran [20,24,30]. The burden of pertussis among infants is high [31]. According to the results of this current study, most of the suspected cases of pertussis were recorded in those cases who were < 1 year similar to a study conducted in Taiwan [32]. The highest number of suspected cases of pertussis was reported in the Spring and the lowest number was reported in the Autumn. The mean (SD) number of suspected cases was 3.46 (2.86) cases a day. Each year, most of the cases were reported from the 21st April to the 21st June, and the lowest cases were reported from the 23rd September to the 22nd October. The highest incidence of pertussis cases was reported in August in the USA in a study between 1990 to 2003 [33]. In another study in Korea, the highest incidence of non-confirmed cases was registered between March and June [34]. The results of a study in Iran showed that the highest cases of pertussis were reported during the Winter and Spring [11]. Other studies in Italy and Australia showed that the highest and lowest incidence of pertussis was seen in the Spring/Summer and in the Autumn/Winter, respectively [35]. Furthermore, the peak of the number of pertussis cases in Australia was seen in the Summer [36]. The seasonal pattern of many infectious diseases is poorly understood. Possible factors include changes in environmental factors that favor conditions for growth, and survival of the microorganisms in the environment, or maybe changes in host behaviors such as going to overcrowded places, thereby allowing contact with other people facilitating pathogen transmission [37]. Factors such as return to school and aggregation of susceptible students in schools appear to be important in the seasonal trend of pertussis incidence [16]. Although, some studies have reported no significant association between school reopening and seasonal trends of pertussis infection [34]. Fluctuations in the incidence of reported cases can also be due to the transmission of pertussis across age groups [11].
- By applying the EWMA estimation method to determine the upper threshold number of pertussis cases that would constitute an outbreak to the national surveillance system data, the “alarm threshold” for pertussis outbreaks, indicated there was 7.55 cases per day, per 10,000,000 people. It means that if the number of reported suspected cases at the national level exceeded 8 cases per day, it should be regarded as an alarm. The EWMA estimation model performed well in the timely detection of influenza outbreaks [30,38].
- The Poisson regression method, estimated the overall “alarm threshold” was 7.50 (4.48–11.06) cases per day, per 10,000,000 people. It means that the upper threshold, the “alarm threshold” is 8 cases per day, but with a conservative approach, 5 cases per day can also be considered in the range of the “alarm threshold.” The Poisson regression method of estimation was reported to have had the best performance in aberration detection of high-count data [39]. The estimated “alarm threshold” by the 2 methods was affected by the number of registered cases. A higher number of diagnosed cases can increase the “alarm threshold” therefore, the estimated level for alarm is in accordance with the current sensitivity of the surveillance system, and can be changed in the future. In other words, improvement of sensitivity of the surveillance system or improvement of the number of reported cases by physicians or healthcare workers will potentially increase the “alarm threshold” in the future. Similarly, the detected aberration using these thresholds can be affected by the performance of physicians and health care workers when it is not a real outbreak. According to the results of both the EWMA and the Poisson regression methods, the “alarm thresholds” for Summer and Autumn were less than the rest of the year. The difference of “alarm threshold” between seasons was affected by the seasonal incidence of reported cases. Due to the lower incidence of pertussis cases in Summer and Autumn, the “alarm threshold” for these seasons was typically lower than the other seasons. This finding was also observed across the strata. The estimated “alarm threshold” in each stratum was affected by the stratum-specific population size. For example, the estimated “alarm threshold” in the second stratum with lower incidence was higher than the first stratum with higher incidence, due to the higher population size. This means that the estimated “alarm threshold” in the second stratum was per 43 million people, while the “alarm threshold” in the first stratum was estimated per 7 million people. After adjusting population size and calculation of the “alarm threshold” per 10 million people, the “alarm threshold” in the first stratum was higher than the other strata. It should be noted that the different incidence proportions and consequently the different threshold levels, cannot be due to the vaccination coverage. This is because the level of vaccination coverage in all provinces of Iran was similar at more than 95% [40] (the population immunity level in Iran was acceptable). In a population without herd immunity or low coverage of vaccine programs, outbreaks of transmissible diseases are inevitable. So, increasing the population immunity level or reaching herd immunity can prevent infectious disease epidemics and outbreaks. By considering high vaccine coverage in Iran, determination of the level of the “alarm threshold” of aberrations may be very helpful for health policymakers for the management of the outbreak, interventions, and efficacy and effectiveness of vaccination programs. Early warning of outbreaks can lead to prophylactic action for at risk people and prevention of outbreaks across the country. Aberrations from the normal trend may be due to wanes in vaccine protection over time, so the health policymakers may implement the use of additional booster doses of pertussis vaccines in adolescents and adults to reduce the number of incident cases to prevent the transmission of infection to infants. Therefore, having information about the numbers of cases indicating “alarm threshold” during each season may play an important role in the timely detection of outbreaks. Policymakers and public health practitioners can make timely decisions to control for possible outbreaks with having information about the possible “alarm thresholds.” These decisions may include forming an outbreak investigation team, and taking steps to confirm the diagnosis, and confirm the existence of an outbreak or epidemic.
- The current study was limited by the sensitivity of the surveillance system which was not the same in different provinces of Iran, so the validity of the estimated upper thresholds by using 2 methods depended on the quality of the registered data. Therefore, underestimation of the actual number of cases by the surveillance system may have influenced the estimations. In addition, there was no clear target or baseline period to calculate the upper control limit, so the researcher determined this period according to available data. Moreover, the data was incomplete on confirmed cases of pertussis so, the data of suspected cases of the pertussis were used. Considering these limitations, the determined “alarm threshold” in the present study may be useful in the early warning of aberrations.
Discussion
- The EWMA and the Poisson regression methods may be used to estimate “alarm thresholds” for an outbreak or early detection of aberrations from the normal trend of infectious diseases. It should be considered that the estimated thresholds may change over time because of the improvement in the sensitivity of the surveillance system and the increasing number of registered cases. Hence, refining the definition of the upper threshold or “alarm threshold” periodically, by considering the sensitivity of the surveillance system, would be warranted. Furthermore, it is recommended that besides using more than 1 method of estimation of upper thresholds, other methods are used to determine the level of “alarm threshold” of pertussis outbreaks in Iran and other countries.
Conclusion
-
Acknowledgements
- The authors would like to express their appreciation for the Iranian Ministry of Health and Center for Communicable Diseases Control for their constant support and collaboration. This study approved by the ethical committee of Tehran University of Medical Sciences with ID:IR.TUMS.SPH.REC.1397.276. This article was extracted from the Ph.D. thesis by Yousef Alimohamadi and financially supported by Tehran University of Medical Sciences.
Acknowledgments
- 1. Kwon HJ, Han SB, Kim BR, et al. Assessment of safety and efficacy against Bordetella pertussis of a new tetanus-reduced dose diphtheria-acellular pertussis vaccine in a murine model. BMC Infect Dis 2017;17:247PMID: 10.1186/s12879-017-2369-x. PMID: 28376777. PMID: 5381055.ArticlePubMedPMCPDF
- 2. Brugueras S, Rius C, Millet J-P, et al. Does the economic recession influence the incidence of pertussis in a cosmopolitan European city? BMC Public Health 2019;19:144PMID: 10.1186/s12889-019-6448-3. PMID: 30717741. PMID: 6360796.ArticlePubMedPMCPDF
- 3. Hara M, Fukuoka M, Tashiro K, et al. Pertussis outbreak in university students and evaluation of acellular pertussis vaccine effectiveness in Japan. BMC Infect Dis 2015;15:45PMID: 10.1186/s12879-015-0777-3. PMID: 25656486. PMID: 4323135.ArticlePubMedPMCPDF
- 4. Nicolai A, Nenna R, Stefanelli P, et al. Bordetella pertussis in infants hospitalized for acute respiratory symptoms remains a concern. BMC Infect Dis 2013;13:526PMID: 10.1186/1471-2334-13-526. PMID: 24209790. PMID: 4226035.ArticlePubMedPMCPDF
- 5. Liu XC, Bell CA, Simmonds KA, et al. Epidemiology of pertussis in Alberta, Canada 2004–2015. BMC Public Health 2017;17:539PMID: 10.1186/s12889-017-4468-4.ArticlePubMedPMC
- 6. Donnan EJ, Fielding JE, Rowe SL, et al. A cross sectional survey of attitudes, awareness and uptake of the parental pertussis booster vaccine as part of a cocooning strategy, Victoria, Australia. BMC Public Health 2013;13:676PMID: 10.1186/1471-2458-13-676. PMID: 23875762. PMID: 3726505.ArticlePubMedPMCPDF
- 7. Böhme M, Voigt K, Balogh E, et al. Pertussis vaccination status and vaccine acceptance among medical students: multicenter study in Germany and Hungary. BMC Public Health 2019;19:182PMID: 10.1186/s12889-019-6516-8. PMID: 30755203. PMID: 6373007.ArticlePubMedPMCPDF
- 8. Fathima S, Ferrato C, Lee BE, et al. Bordetella pertussis in sporadic and outbreak settings in Alberta, Canada, July 2004–December 2012. BMC Infect Dis 2014;14:48PMID: 10.1186/1471-2334-14-48.ArticlePubMedPMC
- 9. Liu X, Wang Z, Zhang J, et al. Pertussis outbreak in a primary school in China: infection and transmission of the macrolide-resistant Bordetella pertussis. Pediatr Infect Dis J 2018;37(6). e145−8. PMID: 10.1097/INF.0000000000001814.ArticlePubMed
- 10. Solano R, Masa-Calles J, Garib Z, et al. Epidemiology of pertussis in two Ibero-American countries with different vaccination policies: lessons derived from different surveillance systems. BMC Public Health 2016;16:1178PMID: 10.1186/s12889-016-3844-9. PMID: 27876021. PMID: 5118903.ArticlePubMedPMCPDF
- 11. Ghorbani GR, Zahraei SM, Moosazadeh M, et al. Comparing seasonal pattern of laboratory confirmed cases of pertussis with clinically suspected cases. Osong Public Health Res Perspect 2016;7(2). 131−7. PMID: 10.1016/j.phrp.2016.02.004. PMID: 27169013. PMID: 4850371.ArticlePubMedPMC
- 12. Zepp F, Heininger U, Mertsola J, et al. Rationale for pertussis booster vaccination throughout life in Europe. Lancet Infect Dis 2011;11(7). 557−70. PMID: 10.1016/S1473-3099(11)70007-X. PMID: 21600850.ArticlePubMed
- 13. Chow MYK, Khandaker G, McIntyre P. Global childhood deaths from pertussis: a historical review. Clin Infect Dis 2016;63(suppl 4). S134−41. PMID: 10.1093/cid/ciw529. PMID: 27838665. PMID: 5106618.ArticlePubMedPMC
- 14. Yeung KHT, Duclos P, Nelson EAS, et al. An update of the global burden of pertussis in children younger than 5 years: a modelling study. Lancet Infect Dis 2017;17(9). 974−80. PMID: 10.1016/S1473-3099(17)30390-0. PMID: 28623146.ArticlePubMed
- 15. Jackson D, Rohani P. Perplexities of pertussis: recent global epidemiological trends and their potential causes. Epidemiol Infect 2014;142(4). 672−84. PMID: 10.1017/S0950268812003093.ArticlePubMed
- 16. de Greeff SC, Dekkers AL, Teunis P, et al. Seasonal patterns in time series of pertussis. Epidemiol Infect 2009;137(10). 1388−95. PMID: 10.1017/S0950268809002489. PMID: 19327200.ArticlePubMed
- 17. Broutin H, Guégan J-F, Elguero E, et al. Large-scale comparative analysis of pertussis population dynamics: periodicity, synchrony, and impact of vaccination. Am J Epidemiol 2005;161(12). 1159−67. PMID: 10.1093/aje/kwi141. PMID: 15937025.ArticlePubMedPDF
- 18. Jõgi P, Oona M, Toompere K, et al. Seroprevalence of IgG antibodies to pertussis toxin in children and adolescents in Estonia. Vaccine 2014;32(41). 5311−5. PMID: 10.1016/j.vaccine.2014.07.066. PMID: 25093282.ArticlePubMed
- 19. Khazaei S, Ayubi E, Mansori K, et al. Pertussis incidence by time, province and age group in Iran, 2006–2011. Iran J Public Health 2016;45(11). 1525−7. PMID: 28028509. PMID: 5182268.PubMedPMC
- 20. Faryadres M, Karami M, Moghimbeigi A, et al. Levels of alarm thresholds of meningitis outbreaks in Hamadan Province, west of Iran. J Res Health Sci 2015;15(1). 62−5. PMID: 25821029.PubMed
- 21. Kleinbaum DG, Kupper LL, Nizam A, et al. Applied regression analysis and other multivariable methods. Nelson Education; 2013.
- 22. Nikbin VS, Shahcheraghi F, Lotfi MN, et al. Comparison of culture and real-time PCR for detection of Bordetella pertussis isolated from patients in Iran. Iran J Microbiol 2013;5(3). 209−14.PubMedPMC
- 23. Templeton KE, Scheltinga SA, van der Zee A, et al. Evaluation of real-time PCR for detection of and discrimination between Bordetella pertussis, Bordetella parapertussis, and Bordetella holmesii for clinical diagnosis. J Clin Microbiol 2003;41(9). 4121−6. PMID: 10.1128/JCM.41.9.4121-4126.2003. PMID: 12958235. PMID: 193783.ArticlePubMedPMC
- 24. Karami M, Soori H, Mehrabi Y, et al. Real time detection of a measles outbreak using the exponentially weighted moving average: does it work? J Res Health Sci 2012;12(1). 25−30. PMID: 22888711.PubMed
- 25. Brookmeyer R, Stroup DF. Monitoring the health of populations: statistical principles and methods for public health surveillance. Oxford (UK): Oxford University Press; 2004.
- 26. Goldenberg A, Shmueli G, Caruana RA, et al. Early statistical detection of anthrax outbreaks by tracking over-the-counter medication sales. Proc Natl Acad Sci U S A 2002;99(8). 5237−40. PMID: 10.1073/pnas.042117499. PMID: 11959973. PMID: 122753.ArticlePubMedPMC
- 27. Farrington C, Andrews NJ, Beale A, et al. A statistical algorithm for the early detection of outbreaks of infectious disease. J R Stat Soc Ser A Stat Soc 1996;159(3). 547−63. PMID: 10.2307/2983331.Article
- 28. Randrianasolo L, Raoelina Y, Ratsitorahina M, et al. Sentinel surveillance system for early outbreak detection in Madagascar. BMC Public Health 2010;10:31PMID: 10.1186/1471-2458-10-31. PMID: 20092624. PMID: 2823701.ArticlePubMedPMCPDF
- 29. Wong W-K, Moore AW, Cooper GF, et al. Bayesian network anomaly pattern detection for disease outbreaks. In: Proceedings of the 20th International Conference on Machine Learning (ICML-03); 2003; 2003; p. 808−15.
- 30. Solgi M, Karami M, Poorolajal J. Timely detection of influenza outbreaks in Iran: Evaluating the performance of the exponentially weighted moving average. J Infect Public Health 2018;11(3). 389−92. PMID: 10.1016/j.jiph.2017.09.011.ArticlePubMed
- 31. Lin Y-C, Yao S-M, Yan J-J, et al. Epidemiological shift in the prevalence of pertussis in Taiwan: implications for pertussis vaccination. J Med Microbiol 2007;56(4). 533−7. PMID: 10.1099/jmm.0.46741-0. PMID: 17374896.ArticlePubMed
- 32. Chang I-F, Lee P-I, Lu C-Y, et al. Resurgence of pertussis in Taiwan during 2009–2015 and its impact on infants. J Microbiol Immunol Infect 2019;52(4). 542−8. PMID: 10.1016/j.jmii.2019.06.002. PMID: 31285158.ArticlePubMed
- 33. Shah AP, Smolensky MH, Burau KD, et al. Seasonality of primarily childhood and young adult infectious diseases in the United States. Chronobiol Int 2006;23(5). 1065−82. PMID: 10.1080/07420520600920718. PMID: 17050218.ArticlePubMed
- 34. Park S, Lee SH, Seo K-H, et al. Epidemiological aspects of pertussis among adults and adolescents in a Korean outpatient setting: a multicenter, PCR-based study. J Korean Med Sci 2014;29(9). 1232−9. PMID: 10.3346/jkms.2014.29.9.1232. PMID: 25246741. PMID: 4168176.ArticlePubMedPMC
- 35. Gonfiantini M, Carloni E, Gesualdo F, et al. Epidemiology of pertussis in Italy: disease trends over the last century. Euro Surveill 2014;19(40). 20921PMID: 10.2807/1560-7917.ES2014.19.40.20921. PMID: 25323077.ArticlePubMed
- 36. Kaczmarek MC, Ware RS, Nimmo GR, et al. Pertussis seasonality evident in polymerase chain reaction and serological testing data, Queensland, Australia. J Pediatric Infect Dis Soc 2015;5(2). 214−7. PMID: 10.1093/jpids/piu144.ArticlePubMedPDF
- 37. Bhatti MM, Rucinski SL, Schwab JJ, et al. Eight-year review of Bordetella pertussis testing reveals seasonal pattern in the United States. J Pediatric Infect Dis Soc 2017;6(1). 91−3.ArticlePubMed
- 38. Steiner SH, Grant K, Coory M, et al. Detecting the start of an influenza outbreak using exponentially weighted moving average charts. BMC Med Inform Decis Mak 2010;10:37PMID: 10.1186/1472-6947-10-37. PMID: 20587013. PMID: 2909986.ArticlePubMedPMCPDF
- 39. Zhou H, Burkom H, Winston CA, et al. Practical comparison of aberration detection algorithms for biosurveillance systems. J Biomed Inform 2015;57:446−55. PMID: 10.1016/j.jbi.2015.08.023. PMID: 26334478.ArticlePubMed
- 40. World Health Organization Iran (Islamic Republic of) [Internet]. WHO and UNICEF estimates of immunization coverage 2018 Available from: https://www.who.int/immunization/monitoring_surveillance/data/irn.pdf?ua=1.
References
Figure 1(A) The suspected cases of pertussis in Iran according to month and (B) the average of pentavalent (Pent3) vaccine coverage in different provinces of Iran in 2017.


Figure 2Line plot of reported cases of suspected pertussis (values) from 25 February 2012 to 23 March 2018 and corresponding levels of overall alarm threshold according to (A) EWMA algorithm and (B) Poisson regression algorithm (red lines).
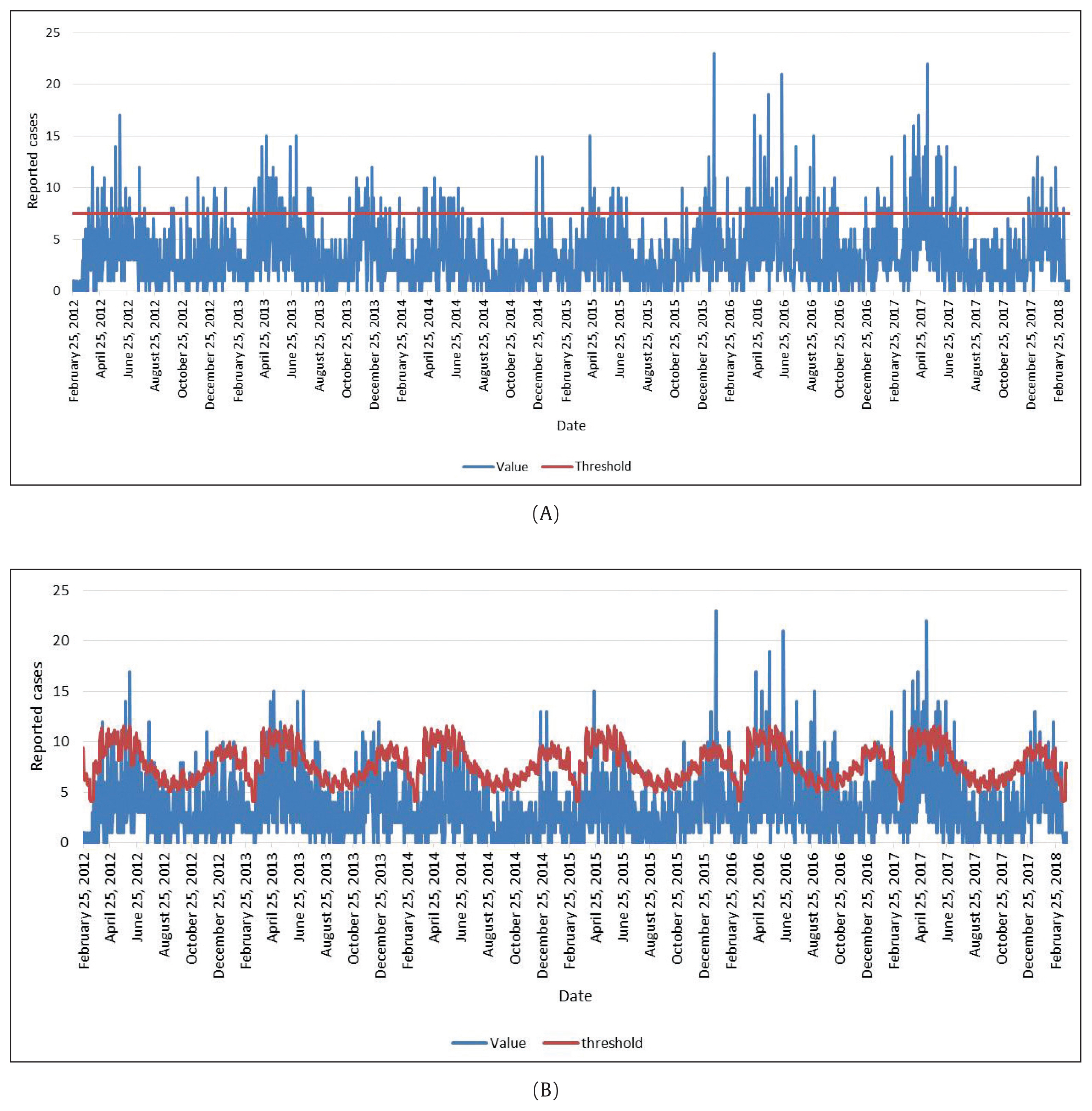

Table 1Descriptive statistics of suspected and confirmed cases of pertussis from the 25th February 2012 to the 23rd March 2018 in Iran.
Table 2Estimated levels of “alarm thresholds” of pertussis outbreak at national level in Iran.
| Indices | Poisson median (range) 95% coverage | EWMA* | |
|---|---|---|---|
| Overall | All ages | 7.50 (4.48–11.06) | 7.55 |
| ≤ 1 | 5.59 (3.55–8.22) | 5.23 | |
| > 1 | 3.90 (2.20–6.36) | 3.28 | |
|
|
|||
| Spring | All ages | 9.82 (6.87–11.51) | 9.36 |
| ≤ 1 | 7.04 (4.93–8.80) | 6.22 | |
| > 1 | 4.80 (3.41–6.95) | 4.06 | |
|
|
|||
| Summer | All ages | 7.01 (4.88–11.84) | 6.65 |
| ≤ 1 | 4.50 (3.38–7.14) | 3.85 | |
| > 1 | 4.22 (2.95–6.87) | 3.39 | |
|
|
|||
| Autumn | All ages | 6.49 (5.14–8.37) | 5.90 |
| ≤ 1 | 4.68 (3.45–6.47) | 3.66 | |
| >1 | 3.51 (1.97–4.97) | 2.66 | |
|
|
|||
| Winter | All ages | 8.40 (4.17–10.91) | 7.24 |
| ≤ 1 | 6.51 (4.14–8.29) | 5.66 | |
| > 1 | 3.65 (1.98–5.11) | 2.69 | |
Table 3Estimated levels of “alarm thresholds” of the pertussis outbreak in all age groups in different clusters according to the incidence of suspected cases.
| Stratum | Indices | Poisson median (95% coverage) | Poisson median (per 10,000,000) (95% coverage) | EWMA* | EWMA (per 10,000,000) |
|---|---|---|---|---|---|
|
Stratum 1 Total population: (7,000,000) |
Overall | 3.55 (2.19–5.46) | 5.07 (3.13–7.80) | 2.68 | 3.83 |
| Spring | 4.09 (2.98–5.84) | 5.84 (4.26–8.34) | 3.39 | 4.84 | |
| Summer | 3.38 (2.52–6.10) | 4.83 (3.60–8.71) | 2.62 | 3.74 | |
| Autumn | 3.28 (2.47–4.02) | 4.69 (3.53–5.74) | 2.54 | 3.63 | |
| Winter | 3.56 (2.00–4.77) | 5.09 (2.86–6.81) | 2.56 | 3.66 | |
|
Stratum 2 Total population: (43,000,000) |
Overall | 5.45 (3.50–7.62) | 1.27 (0.81–1.77) | 5.01 | 1.17 |
| Spring | 6.58 (4.94–8.40) | 1.53 (1.15–1.95) | 5.86 | 1.36 | |
| Summer | 4.65 (3.40–7.14) | 1.08 (1.08–1.66) | 4.26 | 0.99 | |
| Autumn | 4.32 (3.22–6.36) | 1.00 (0.75–1.48) | 4.35 | 1.01 | |
| Winter | 6.09 (4.02–7.70) | 1.42 (0.93–1.79) | 5.33 | 1.24 | |
|
Stratum 3 Total population: (30,000,000) |
Overall | 2.01 (1.15–3.11) | 0.67 (0.38–1.04) | 1.08 | 0.36 |
| Spring | 2.51 (1.54–3.52) | 0.84 (0.51–1.17) | 1.67 | 0.56 | |
| Summer | 2.03 (1.18–2.93) | 0.68 (0.39–0.98) | 1.04 | 0.35 | |
| Autumn | 1.77 (1.31–2.31) | 0.59 (0.44–0.77) | 0.99 | 0.33 | |
| Winter | 1.98 (1.05–2.63) | 0.66 (0.35–0.88) | 1.02 | 0.34 |
Figure & Data
References
Citations
Citations to this article as recorded by 

- Immunogenicity and Safety of a Newly Developed Tetanus-Diphtheria Toxoid (Td) in Healthy Korean Adolescents: a Multi-center, Randomized, Double-blind, Active-Controlled Phase 3 Trial
Ui Yoon Choi, Ki Hwan Kim, Jin Lee, Byung Wook Eun, Hwang Min Kim, Kyung-Yil Lee, Dong Ho Kim, Sang Hyuk Ma, Jina Lee, Jong-Hyun Kim
Journal of Korean Medical Science.2021;[Epub] CrossRef


 PubReader
PubReader ePub Link
ePub Link Cite
Cite