Articles
- Page Path
- HOME > Osong Public Health Res Perspect > Volume 8(5); 2017 > Article
-
Original Article
A Retrospective Mid- and Long-term Follow-up Study on the Changes in Hematologic Parameters in the Highly Exposed Residents of the Hebei Spirit Oil Spill in Taean, South Korea - Young-Hyun Choi, Jee-Young Hong, Moo-Sik Lee
-
Osong Public Health and Research Perspectives 2017;8(5):358-366.
DOI: https://doi.org/10.24171/j.phrp.2017.8.5.10
Published online: October 31, 2017
Department of Preventive Medicine and Public Health, College of Medicine, Konyang University, Daejeon, Korea
- Corresponding author: Moo-Sik Lee, E-mail: mslee@konyang.ac.kr
Copyright ©2017, Korea Centers for Disease Control and Prevention
This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
Abstract
-
Objectives
- This study aimed to analyze changes in hematologic parameters in the residents of the areas highly contaminated by the Hebei Spirit Oil Spill in 2007 and those who participated in the clean-up activities.
-
Methods
- According to demographic characteristics, health status and behavior, and level of exposure to oil, we compared the hematologic results in 2009 and 2012 among 701 residents. The hematologic parameters were composed of white blood cell (WBC) count, and levels of hemoglobin, hematocrit (Hct), aspartate aminotransferase (AST), alanine aminotransferase (ALT), glucose, glycosylated hemoglobin (HbA1c), blood urea nitrogen (BUN), creatinine (Cr), total cholesterol (T-chol), high-density lipoprotein (HDL), and triglyceride (TG).
-
Results
- Paired t-test revealed that the WBC count and levels of Hct, AST, ALT, glucose, and HbA1c significantly increased, whereas the BUN, Cr, HDL, and TG levels significantly decreased. Multiple linear regression modelling showed a relationship between the level of exposure to oil and temporal changes in Hct, glucose, HbA1c, and BUN levels.
-
Conclusion
- Our results suggest a relationship between level of exposure to oil and changes in hematologic parameters over 3 years. Further studies should be conducted to determine the impact of oil spill on health such as the occurrence of diseases.
- On December 7, 2007, Hebei Spirit, a Hong Kong-registered oil tanker, was rammed by a crane-carrying barge of Samsung Heavy Industries tugboats. Approximately 10,900 tons of oil spilled into the sea, which contaminated 1,052 km of coastline in Taean county, Korea [1]. This accident, named the Hebei Spirit Oil Spill, was the largest oil spill accident in South Korea.
- The spilled oil contained volatile organic compounds (VOCs), benzene, toluene, ethylbenzene, and xylene (BTEX), polycystic aromatic hydrocarbons (PAHs), and heavy metals [2]. Exposure to these components of crude oil increases the risk of many health problems such as respiratory symptoms, nervous symptoms, dermatologic symptoms, and cancer [3–7]. During the 7 months after the Hebei Spirit Oil Spill, more than 2 million volunteers, including 556,323 Taean residents, participated in clean-up activities [2]. Many participants wore insufficiently appropriate personal protective equipment [8] and reported several health problems, including respiratory, dermatologic, and ophthalmic symptoms [9].
- After oil spill accidents abroad, many studies have been conducted to investigate the health impact such as acute symptoms [6,7,10,11]; mental health, including anxiety, posttraumatic stress syndrome (PTSD), and depression [12,13]; pulmonary function testing [14]; and blood tests, including liver function, renal function, and hematologic profile [15–17].
- Various studies were also conducted to investigate the health impact after the Hebei Spirit Oil Spill [18–20]. However, most of the previous studies on the oil spill incident in Hebei Spirit were cross-sectional studies related to acute physical symptoms or mental health, or exposure to oil. No studies have been conducted on the mid- to long-term effects of oil toxicants, including the effects on hematologic, liver, and renal functions. Therefore, this study aimed to analyze mid- to long-term changes in hematologic and biochemical parameters, and related factors among residents of the highly exposed areas after the Hebei Spirit Oil Spill accident.
INTRODUCTION
- 1. Study subjects
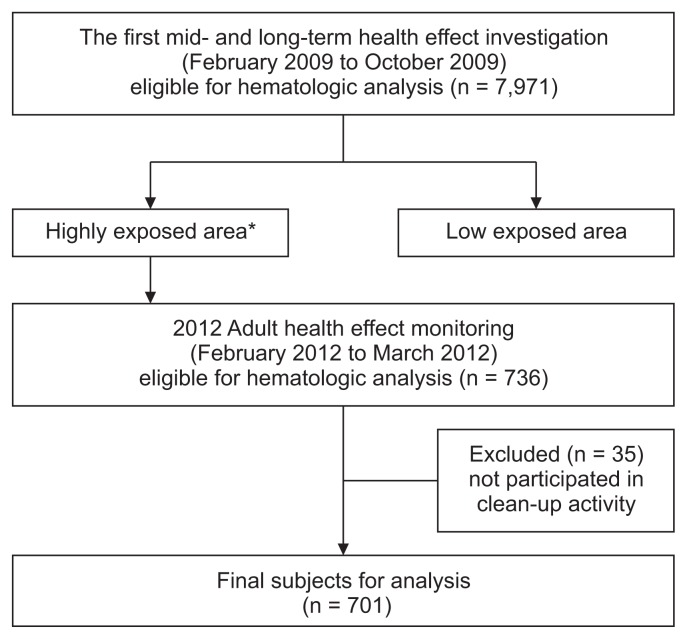
- The study subjects were 701 adults who responded to the first survey and monitoring of health impacts in 2009 and participated in the follow-up monitoring in 2012. The first survey was conducted among 9,246 residents of Taean county from January 2009 to June 2010 by using structured questionnaires. Of these participants, 7,971 underwent blood tests for investigating their hematologic profile, liver function, and renal function. In 2012, a follow-up monitoring was conducted among 891 adults who were living in the area most polluted by the oil spill accident from February to March 2012. Residents living in Sowon-myeon, Wonbuk-myeon, Iwon-myeon, Geunheung-myeon, which were exposed to the coasts contaminated by the thick oil layer at the initial stage of the accident, were classified as highly exposed subjects to oil. Of these residents, 701 adults participated in the first survey and the clean-up work (Figure 1).
- This study was approved by the Dankook University Hospital Institutional Review Board (DKUH201406013-HE003).
- 2. Independent variables
- The independent variables were demographic characteristics, health status and behavioral characteristics, and oil exposure characteristics. Independent variables were classified into three categories, namely demographic, health behavior, and the level of exposure to oil.
- The demographic characteristics included sex, age, educational attainment, marital status, type of health insurance, monthly household income, religion, and occupation. The age variable was divided into age groups of 26–49, 50–59, 60–69, and 70–81 years, respectively. Educational attainment was classified as below elementary school graduation, elementary school graduation, and above middle school graduation. Marital status was classified as married and other. The types of health insurance were national health insurance and Medicaid. Monthly household income was divided into < 500,000, 500,000–999,999, 1,000,000–1,999,999 and > 2 million KRW. Religion was divided into presence or absence of a religion. Occupation was divided into farmer-fisherman and others.
- Health behavior variables included body mass index (BMI), presence of chronic disease, smoking status, drinking status, and frequency of weekly exercise. BMI was divided into three subgroups: normal, overweight, and obese. Smoking was classified into three groups, namely nonsmokers, ex-smokers, and current smokers. The numbers of weekly exercises were divided into 1 or 2 times, 3 or 4 times, and 5 or 6 times.
- The levels of exposure to spilled oil were categorized according to the days of acute-phase clean-up work, the days of total clean-up work, the distance from the accident point to the residence, the distance from the initial pollution shore to the residence, respiratory symptoms, skin symptoms, and risk of posttraumatic stress disorder.
- Days of participating in the clean-up work during the acute phase of the Hebei Spirit Oil Spill accident were divided into 1–14, 15–19, and 20–24 days, from December 7 to 31, 2007. The total numbers of days of participation in the clean-up work up to the date of the first survey were divided into 3–60, 61–90, 91–120, 121–150, and 151–325 days. The distance from the accident site to the residence area were divided into ≤ 14.0, 14.1–15.0, 15.1–16.0, 16.1–17.0, > 17.0 km. The distances from the contaminated seashore to the residence area was measured using the geographic information system and divided into ≤ 0.20, 0.21–0.40, 0.41–0.60, 0.61–0.80, > 0.80 km. The risk of posttraumatic stress disorder was defined as a total score of ≥ 15 in the 17 items of the posttraumatic diagnostic scale [21,22].
- 3. Outcome variables
- Outcome variables included 14 items of blood tests, including complete blood count (CBC), liver function test, renal function test, diabetic mellitus test, and lipid test. The CBC test results were composed of white blood cell (WBC) count, hemoglobin (Hb) level, hematocrit (Hct) level, and platelet (PLT) count. The liver function test included the levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), and gamma glutamyl transferase (γ-GT). Blood urea nitrogen (BUN) and creatinine (Cr) were tested to reflect kidney function. Fasting blood glucose (glucose) and glycosylated hemoglobin (HbA1c) levels were measured. We also tested for total cholesterol (T-chol), high-density lipoprotein (HDL), and triglyceride (TG) levels.
- 4. Statistical analysis
- A paired t-test was conducted to examine differences in measurements in the 14 items of the blood test between years 2009 and 2012. Independent sample t-test, one-way analysis of variance, and linear regression analysis were used to determine any differences in the levels of the 14 blood test items according to demographic, health behavior, and exposure level to oil. Data were analyzed using IBM SPSS Statistics ver. 20.0 (IBM Co., Armonk, NY, USA) based on a significance level of 0.05.
MATERIALS AND METHODS
- Table 1 shows the demographic characteristics of the study subjects according to sex. The independent variables of educational attainment, marital status, monthly household income, type of occupation, smoking, and drinking showed statically significant differences according to sex. Age, type of health insurance, BMI, presence of chronic disease, and weekly frequency of exercise showed no significant difference according to sex.
- The levels of exposure to oil according to sex are displayed in Table 2. The numbers of days of participating in clean-up work during the acute phase and total clean-up work were significantly higher for men than for women. The distance from the accident site and contaminated seashore, the presence of respiratory and dermatologic symptoms, and the risk of PTSD showed no significant differences by sex.
- We compared blood test results between 2009 and 2012 (Table 3) and found statistically differences in WBC count and levels of Hct, AST, ALT, BUN, Cr, glucose, HbA1c, HDL, and TG. The test results for WBC count and levels of Hct, AST, ALT, glucose, and HbA1c in 2012 significantly increased from those in 2009, but the test results for BUN, Cr, HDL, and TG levels significantly decreased.
- Linear regression analyses were conducted to find contributing factors to changes in blood test results for WBC count, and levels of Hct, AST, ALT, BUN, Cr, glucose, HbA1c, HDL, and TG between 2009 and 2012 (Table 4). None of the predictive factors affected the difference in WBC count. Long distance from the contaminated seashore to the residence areas significantly contributed to the negative change in Hct level (R2 = 0.018, F = 3.028). High monthly household income was a significant predictor of positive change in AST level (R2 = 0.025, F = 4.426). Residents with Medicaid and higher monthly household income showed a significant relationship to positive change in ALT level (R2 = 0.020, F = 3.549). Long distances from the accident site and contaminated seashore to the residence area were related to negative change in glucose level (R2 = 0.053, F = 4.077). Residents with obesity and without respiratory symptoms were related to negative change in HbA1c level (R2 = 0.021, F = 4.804). BUN level negatively changed in the residents who were elderly men with obesity, participated in total clean-up work during a short period, and had no respiratory symptom and PTSD risk (R2 = 0.071, F = 2.984). Negative changes in HDL level were found in the residents who had no religion, were farmers and fishermen, were ex-smokers, and had no drinking habit (R2 = 0.006, F = 3.078). Residents who had no religion, were farmers and fishermen, were nonsmokers, and had no drinking habit showed positive changes in TG level (R2 = 0.044, F = 6.372). Negative changes in Cr level were observed in the residents who were men and had obesity, and had no chronic disease (R2 = 0.215, F = 13.853).
RESULTS
- The characteristics of the exposure to oil from the Hebei Spirit Oil Spill contributed to the changes in the various blood test results between years 2009 and 2012. Living in the area close to the accident site and contaminated seashore was related to the changes in the test results for Hct, glucose, BUN, and HDL levels. Residents who participated in clean-up work during 2 or more months showed positive changes in BUN level. These results are consistent with the findings of previous studies [15–17] that reported changes in blood, liver, respiratory, kidney, and nervous system functions after an oil exposure accident. Spilled oil that contains various components such as VOCs and PAHs may remain in the environment and negatively impact human health and ecosystems during several years. The higher the pollution level of the area, the higher the participation rate of the residents in the clean-up work, the longer the work period and time, and the higher the possibility of getting oil on clothes, and the face and limbs [19]. When VOCs and PAHs are mainly absorbed through the respiration or the skin [6], these substances are mainly metabolized in the liver. The metabolites are excreted in urine and produce reactive oxygen species that cause oxidative stress in the body, affecting biomaterials such as DNA, protein, and lipids [23].
- Of the oil components that were released, only a few are known to have toxic and adverse health effects. Most toxic components are known to be toxic only when they act as a single substance, and their toxic mechanisms have not been studied in mixtures such as crude oil. In a study of the association of organic solvent and lead exposure to hepatic function tests results, Chang et al. [24] found that the group of workers exposed simultaneously to organic solvents and lead showed a greater increase in liver function index than those exposed to only a single substance. Accordingly, additive actions and interactions should be considered in mixed substance exposure, even if heavy metals such as organic solvents and lead are at or below the permissible exposure limit. If each substance is exposed to the same or lower than the permitted exposure standard, it may be harmful to the human body [25].
- The limitations of this study are as follows: The blood test results used in this study were not the result of an acute period of several days or weeks after the accident but rather the result of a considerable period after the accident, so many confounding factors such as age, personal illness, eating habits, and environment can be interrelated. Therefore, interpretation of the results is limited. Unfortunately, in the case of the largest oil spill incident in the history of the Republic of Korea, both the government and the residents were ignorant about the necessity of a health impact investigation. The residents were forced to ignore the immediate effects of the oil spill on their livelihood, so the right time for the investigation was missed. If this situation is resumed in Korea or abroad, it is desirable to conduct a survey on the health effects of the acute period in addition to the control work. In 2010, the National Institute of Environmental Health Sciences (NIEHS) of the National Institutes of Health conducted a health impact survey in residents during an acute period in a British oil spill accident [25].
- Another limitation was that we conducted this survey only among residents who participated in the oil accident prevention work without a control group. Most previous studies on the health impact of oil spill accidents compared an exposed group with a control group of subjects who were not exposed to the oil accident. This is useful for the generalization of the results of the study because the effect of the oil exposure can be more clearly and easily confirmed by comparing the results of the exposed and unexposed groups when a control group is present. In 2010, the United States Gulf of Mexico British Petroleum oil spill incident was also subjected to a cross-sectional survey led by the NIEHS and a follow-up survey of residents and control workers [15,25]. In environmental health research, a control group with similar characteristics as the exposed group is not easy to obtain, and obtaining consent from the farmers and villagers who are busy in agriculture and fisheries is practically impossible. In some cases, it is difficult to interpret the differences in results between exposure to oil toxins and other personal factors.
- Our results suggest that exposure to spilled oil is a risk factor for worsening changes in some blood test results such as Hct, glucose, BUN, and HDL levels. Further studies should be conducted to demonstrate the causal relationship between oil spill exposure and the incidence or aggravation of diseases such as diabetes mellitus and renal failure, which are related to exposure to VOCs and PAHs in crude oil.
DISCUSSION
-
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
Article information
- 1. Ha M, Lee WJ, Lee S, et al. A literature review on health effects of exposure to oil spill. J Prev Med Public Health 2008;41:345−54. In Korean. https://doi.org/10.3961/jpmph.2008.41.5.345. PMID: 10.3961/jpmph.2008.41.5.345. PMID: 18827503.
- 2. Korean Ocean Research and Development Institute. Data of analysis results of Hebei Spirit crude oil. Ansan: Marine Safety and Pollution Response Research Department, Korean Ocean Research and Development Institute; 2008.
- 3. LaDou J. Current occupational & environmental medicine. 3rd ed. New York: McGraw-Hill; 2004.
- 4. International Agency for Research on Cancer. IARC monographs on the evaluation of carcinogenic risks to humans, vol. 29. Some industrial chemicals and dyestuffs summary of data reported and evaluation. Lyon: International Agency for Research on Cancer; 1982.
- 5. Janjua NZ, Kasi PM, Nawaz H, et al. Acute health effects of the Tasman Spirit oil spill on residents of Karachi, Pakistan. BMC Public Health 2006;6:84https://doi.org/10.1186/1471-2458-6-84. PMID: 10.1186/1471-2458-6-84. PMID: 16584541.ArticlePubMedPMC
- 6. Morita A, Kusaka Y, Deguchi Y, et al. Acute health problems among the people engaged in the cleanup of the Nakhodka oil spill. Environ Res 1999;81:185−94. https://doi.org/10.1006/enrs.1999.3979. PMID: 10.1006/enrs.1999.3979. PMID: 10585014.ArticlePubMed
- 7. Lyons RA, Temple JM, Evans D, et al. Acute health effects of the Sea Empress oil spill. J Epidemiol Community Health 1999;53:306−10. https://doi.org/10.1136/jech.53.5.306. PMID: 10.1136/jech.53.5.306. PMID: 10396538.ArticlePubMedPMC
- 8. Kim YM, Cheong HK, Kim JH, et al. Scientific basis of environmental health contingency planning for a coastal oil spill. J Prev Med Public Health 2009;42:73−81. In Korean. https://doi.org/10.3961/jpmph.2009.42.2.73. PMID: 10.3961/jpmph.2009.42.2.73. PMID: 19349735.
- 9. Lee CH, Kang YA, Chang KJ, et al. Acute health effects of the Hebei oil spill on the residents of Taean, Korea. J Prev Med Public Health 2010;43:166−73. https://doi.org/10.3961/jpmph.2010.43.2.166. PMID: 10.3961/jpmph.2010.43.2.166. PMID: 20383050.ArticlePubMed
- 10. Campbell D, Cox D, Crum J, et al. Initial effects of the grounding of the tanker Braer on health in Shetland. The Shetland Health Study Group. BMJ 1993;307:1251−5. https://doi.org/10.1136/bmj.307.6914.1251. PMID: 10.1136/bmj.307.6914.1251. PMID: 8281057.ArticlePubMedPMC
- 11. Campbell D, Cox D, Crum J, et al. Later effects of grounding of tanker Braer on health in Shetland. BMJ 1994;309:773−4. https://doi.org/10.1136/bmj.309.6957.773. PMID: 10.1136/bmj.309.6957.773. PMID: 7950562.ArticlePubMedPMC
- 12. Palinkas LA, Petterson JS, Russell J, et al. Community patterns of psychiatric disorders after the Exxon Valdez oil spill. Am J Psychiatry 1993;150:1517−23. https://doi.org/10.1176/ajp.150.10.1517. PMID: 10.1176/ajp.150.10.1517. PMID: 8379557.ArticlePubMed
- 13. Gallacher J, Bronstering K, Palmer S, et al. Symptomatology attributable to psychological exposure to a chemical incident: a natural experiment. J Epidemiol Community Health 2007;61:506−12. https://doi.org/10.1136/jech.2006.046987. PMID: 10.1136/jech.2006.046987. PMID: 17496259.ArticlePubMedPMC
- 14. Crum JE. Peak expiratory flow rate in schoolchildren living close to Braer oil spill. BMJ 1993;307:23−4. PMID: 10.1136/bmj.307.6895.23. PMID: 8343663.ArticlePubMedPMC
- 15. Goldstein BD, Osofsky HJ, Lichtveld MY. The Gulf oil spill. N Engl J Med 2011;364:1334−48. https://doi.org/10.1056/NEJMra1007197. PMID: 10.1056/NEJMra1007197. PMID: 21470011.ArticlePubMed
- 16. Diaz JH. The legacy of the Gulf oil spill: analyzing acute public health effects and predicting chronic ones in Louisiana. Am J Disaster Med 2011;6:5−22. PMID: 21466025.ArticlePubMed
- 17. D’Andrea MA, Reddy GK. Health consequences among subjects involved in Gulf oil spill clean-up activities. Am J Med 2013;126:966−74. https://doi.org/10.1016/j.amjmed.2013.05.014. PMID: 10.1016/j.amjmed.2013.05.014. PMID: 24050487.ArticlePubMed
- 18. Gwack J, Lee JH, Kang YA, et al. Acute health effects among military personnel participating in the cleanup of the hebei spirit oil spill, 2007, in taean county, Korea. Osong Public Health Res Perspect 2012;3:206−12. https://doi.org/10.1016/j.phrp.2012.10.001. PMID: 10.1016/j.phrp.2012.10.001. PMID: 24159516.ArticlePubMedPMC
- 19. Ha M. Investigation of acute health problems among people exposed to the Hebei Spirit oil spill. Seoul: Ministry of Environment; 2008.
- 20. Lee KH, Park SY, Ahn JW, et al. A study of air pollution due to oil spill accident at the Tae-Ahn Peninsula, Korea 2007 da major focus on hydrocarbon pollution. J Korean Soc Odor Res Eng 2008;7:68−75.
- 21. Foa EB, Cashman L, Jaycox L, et al. The validation of a self-report measure of posttraumatic stress disorder: The Posttraumatic Diagnostic Scale. Psychol Assess 1997;9:445−51. https://doi.org/10.1037/1040-3590.9.4.445. PMID: 10.1037/1040-3590.9.4.445.Article
- 22. Nam BR, Kwon HI, Kwon JH. Psychometric qualities of the Korean version of the Posttraumatic Diagnosis Scale(PDS-K). Korean J Clin Psychol 2010;29:147−67. PMID: 10.15842/kjcp.2010.29.1.009.Article
- 23. Fridovich I. The biology of oxygen radicals. Science 1978;201:875−80. https://doi.org/10.1126/science.210504. PMID: 10.1126/science.210504. PMID: 210504.ArticlePubMed
- 24. Chang WJ, Joe KT, Park HY, et al. The relationship of liver function tests to mixed exposure to lead and organic solvents. Ann Occup Environ Med 2013;25:5https://doi.org/10.1186/2052-4374-25-5. PMID: 10.1186/2052-4374-25-5. PMID: 24472152.ArticlePubMedPMC
- 25. National Institute of Environmental Health Sciences (NIEHS). Gulf long-term follow-up study. Research Triangle Park: NIEHS; 2011.
REFERENCES
| Variable | Male | Female | Total |
|---|---|---|---|
| Age (y) | |||
| 26–49 | 24 (9.2) | 47 (10.7) | 71 (10.1) |
| 50–59 | 61 (23.5) | 119 (27.0) | 180 (25.7) |
| 60–69 | 104 (40.0) | 181 (41.0) | 285 (40.7) |
| 70–81 | 71 (27.3) | 94 (21.3) | 165 (23.5) |
| Educational attainmenta | |||
| Kindergarten or lower | 30 (11.5) | 227 (51.5) | 257 (36.7) |
| Elementary school | 128 (49.2) | 152 (34.5) | 280 (39.9) |
| Middle school or higher | 102 (39.2) | 62 (14.1) | 164 (23.4) |
| Marital statusa | |||
| Married | 242 (93.1) | 329 (74.6) | 571 (81.5) |
| Others | 18 (6.9) | 112 (25.4) | 130 (18.5) |
| Type of health insurance | |||
| National health insurance | 154 (59.2) | 258 (58.5) | 412 (58.8) |
| Medicaid | 106 (40.8) | 183 (41.5) | 289 (41.2) |
| Monthly household income (KRW)a | |||
| < 500,000 | 144 (55.4) | 293 (66.4) | 437 (62.3) |
| 500,000–999,999 | 35 (13.5) | 67 (15.2) | 102 (14.6) |
| 1,000,000–1,999,999 | 49 (18.8) | 51 (11.6) | 100 (14.3) |
| ≥ 2,000,000 | 32 (12.3) | 30 (6.8) | 62 (8.8) |
| Religiona | |||
| Yes | 120 (46.2) | 257 (58.3) | 377 (53.8) |
| No | 140 (53.8) | 184 (41.7) | 324 (46.2) |
| Occupationa | |||
| Farming and fishing | 154 (59.2) | 182 (41.3) | 336 (47.9) |
| Others | 106 (40.8) | 259 (58.7) | 365 (52.1) |
| Body mass index (kg/m2) | |||
| Underweight to normal (< 23.0) | 77 (30.8) | 119 (27.8) | 196 (28.9) |
| Overweight (23.0–25.0) | 61 (24.4) | 101 (23.6) | 162 (23.9) |
| Obese (> 25.0) | 112 (44.8) | 208 (48.6) | 320 (47.2) |
| Chronic diseaseb | |||
| Yes | 112 (43.1) | 190 (43.1) | 302 (43.1) |
| No | 148 (56.9) | 251 (56.9) | 399 (56.9) |
| Smokinga | |||
| Non-smoker | 140 (54.3) | 428 (97.3) | 568 (81.4) |
| Ex-smoker | 61 (23.6) | 4 (0.9) | 65 (9.3) |
| Smoker | 57 (22.1) | 8 (1.8) | 65 (9.3) |
| Drinkinga | |||
| Yes | 139 (53.9) | 54 (12.3) | 193 (27.7) |
| No | 119 (46.1) | 386 (87.7) | 505 (72.3) |
| Weekly exercise frequency | |||
| None | 181 (70.2) | 313 (71.1) | 494 (70.8) |
| 1 or 2 times | 13 (5.0) | 37 (8.4) | 50 (7.2) |
| 3 or 4 times | 24 (9.3) | 36 (8.2) | 60 (8.6) |
| 5 or 6 times | 40 (15.5) | 54 (12.3) | 94 (13.5) |
| Total | 260 (100.0) | 441 (100.0) | 701 (100.0) |
Values are presented as number (%).
Because study subjects didn’t respond to some independent variables, sum of frequencies in this table are less than number of total subjects.
a p < 0.05 by chi-square test.
b Chronic diseases: hypertension, diabetes mellitus, asthma, thyroid disease, anemia, chronic obstructive pulmonary disease, stroke and cerebrovascular disease, cardiac angina, and cardiovascular disease.
| Variable | Male | Female | Total |
|---|---|---|---|
| Number of clean-up days in acute stagea | |||
| 1–14 | 10 (3.7) | 32 (6.9) | 42 (5.7) |
| 15–19 | 14 (5.5) | 37 (8.6) | 51 (7.5) |
| 20–24 | 232 (90.6) | 359 (83.9) | 591 (86.4) |
| Total number of clean-up daysa | |||
| 3–60 | 26 (10.0) | 76 (17.3) | 102 (14.6) |
| 61–90 | 30 (11.6) | 83 (18.9) | 113 (16.2) |
| 91–120 | 41 (15.8) | 70 (15.9) | 111 (15.9) |
| 121–150 | 84 (32.4) | 125 (28.5) | 209 (29.9) |
| 151–325 | 78 (30.1) | 85 (19.4) | 163 (23.4) |
| Distance from the accident point (km) | |||
| ≤ 14.0 | 48 (18.7) | 81 (18.5) | 129 (18.6) |
| 14.1–15.0 | 68 (26.5) | 92 (21.1) | 160 (23.1) |
| 15.1–16.0 | 62 (24.1) | 106 (24.3) | 168 (24.2) |
| 16.1–17.0 | 21 (8.2) | 54 (12.4) | 75 (10.8) |
| > 17.0 | 58 (22.6) | 104 (23.8) | 162 (23.3) |
| Distance from contaminated seashore (km) | |||
| ≤ 0.20 | 56 (22.3) | 91 (21.5) | 147 (21.8) |
| 0.21–0.40 | 38 (15.1) | 65 (15.3) | 103 (15.3) |
| 0.41–0.60 | 56 (22.3) | 92 (21.7) | 148 (21.9) |
| 0.61–0.80 | 40 (15.9) | 60 (14.2) | 100 (14.8) |
| > 0.80 | 61 (24.3) | 116 (27.4) | 177 (26.2) |
| Respiratory symptoms | |||
| Yes | 122 (47.1) | 203 (46.1) | 325 (46.5) |
| No | 137 (52.9) | 237 (53.9) | 374 (53.5) |
| Dermatologic symptoms | |||
| Yes | 53 (20.5) | 108 (24.5) | 161 (23.0) |
| No | 206 (79.5) | 332 (75.5) | 538 (77.0) |
| PTSD risk | |||
| Yes | 87 (33.9) | 173 (40.0) | 260 (37.7) |
| No | 170 (66.1) | 260 (60.0) | 430 (62.3) |
| Total | 273 (100.0) | 463 (100.0) | 736 (100.0) |
Values are presented as number (%).
Because study subjects didn’t respond to some independent variables, sum of frequencies in this table are less than number of total subjects.
Acute stage: December 7–31, 2007; Respiratory symptoms: wheezing, sneezing, rhinorrhea, or allergic rhinitis during living or exercising in the recent 12 months; Dermatologic symptoms: eczema, urticaria, or atopy in the recent 12 months; PTSD risk: a score of ≥ 15 points in the posttraumatic diagnostic scale questionnaire.
PTSD: posttraumatic stress syndrome.
a p < 0.05 by chi-square test.
| Hematologic tests | Subject (n) | 2009 | 2012 | p-valuea |
|---|---|---|---|---|
| CBC | ||||
| WBC count (/μL) | 698 | 6,176.5 ± 1672.9 | 6,356.0 ± 2034.1 | 0.011 |
| Hb (mg/dL) | 698 | 14.0 ± 1.3 | 14.0 ± 1.3 | 0.798 |
| Hct (%) | 698 | 39.3 ± 3.5 | 39.5 ± 3.5 | 0.027 |
| PLT count (/μL) | 692 | 25.1 ± 5.3 | 25.2 ± 6.1 | 0.532 |
| Biochemistry | ||||
| AST (IU/L) | 699 | 26.7 ± 13.5 | 29.0 ± 12.3 | 0.000 |
| ALT (IU/L) | 699 | 25.8 ± 16.9 | 28.2 ± 18.9 | 0.002 |
| γ-GT (IU/L) | 699 | 44.7 ± 61.4 | 41.7 ± 55.4 | 0.143 |
| Glucose (mg/dL) | 699 | 103.6 ± 22.8 | 106.6 ± 25.0 | 0.000 |
| HbA1c (%) | 698 | 5.9 ± 0.7 | 6.1 ± 0.8 | 0.000 |
| BUN (mg/dL) | 699 | 16.1 ± 4.7 | 15.3 ± 4.1 | 0.001 |
| Cr (mg/dL) | 699 | 0.9 ± 0.2 | 0.8 ± 0.2 | 0.000 |
| T-chol (mg/dL) | 699 | 207.9 ± 39.8 | 206.2 ± 38.3 | 0.250 |
| HDL (mg/dL) | 699 | 51.9 ± 12.2 | 50.1 ± 12.3 | 0.000 |
| TG (mg/dL) | 699 | 145.9 ± 99.9 | 135.4 ± 93.1 | 0.004 |
Values are presented as mean ± standard deviation.
CBC, complete blood count; WBC, white blood cell; Hb, hemoglobin; Hct, hematocrit; PLT, platelet; AST, aspartate aminotransferase; ALT, alanine aminotransferase; γ-GT, gamma glutamyl transferase; HbA1c, glycosylated hemoglobin; BUN, blood urea nitrogen; Cr, creatinine; T-chol, total cholesterol; HDL, high-density lipoprotein; TG, triglyceride.
a By paired t-test.
| Variable | Regression coefficients | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Hct | AST | ALT | Glucose | HbA1c | BUN | HDL | TG | Cr | |
| Women (ref. men) | 1.089* | 0.215* | |||||||
| Age (y) (ref. 26–49 y) | |||||||||
| 50–59 | −0.941 | ||||||||
| 60–69 | −1.444 | ||||||||
| 70–81 | −2.514* | ||||||||
| Education (ref. kindergarten or lower) | |||||||||
| Elementary school | 0.000 | ||||||||
| Middle school or higher | −0.012 | ||||||||
| Marital status (ref. married) | −2.498 | ||||||||
| Medicaid (ref. health insurance) | 3.686* | 2.621 | |||||||
| Monthly household income (KRW) (ref. < 500,000 KRW) | |||||||||
| 500,000–999,999 | 5.372* | 4.879* | 0.016 | ||||||
| 1,000,000–1,999,999 | 3.653* | 4.447* | −0.017 | ||||||
| ≥ 2,000,000 | 0.029 | −0.480 | −0.058 | ||||||
| Not having a religion (ref. yes) | 19.108* | −0.028 | |||||||
| Occupation (ref. farming and fishing) | −15.888* | ||||||||
| Body mass index (kg/m2) (ref. < 23.0 kg/m2) | |||||||||
| 23.0–25.0 | −0.004 | −1.330 | −0.038 | ||||||
| > 25.0 | −0.120* | −1.726* | −0.063* | ||||||
| Presence of a chronic disease (ref. no) | −0.051* | ||||||||
| Smoking (ref. nonsmoker) | |||||||||
| Ex-smoker | 0.478 | −36.379* | −0.036 | ||||||
| Smoker | −3.093* | 14.253 | −0.044 | ||||||
| Drinking (ref. none) | 17.321* | 0.026 | |||||||
| Number of days of clean-up work during the acute stage (ref. 1–14) | |||||||||
| 15–19 | −10.584* | ||||||||
| 20–24 | −12.226* | ||||||||
| Number of days of total clean-up work (ref. 3–60) | |||||||||
| 61–90 | 1.435 | ||||||||
| 91–120 | −0.897 | ||||||||
| 121–150 | 0.035 | ||||||||
| 151–325 | 0.739 | ||||||||
| Distance (km) from the accident site (ref. > 17.0) | |||||||||
| ≤ 14.0 | −6.384* | −3.225* | |||||||
| 14.1–15.0 | 0.120 | 0.373* | |||||||
| 15.1–16.0 | 2.012 | −2.207* | |||||||
| 16.1–17.0 | 0.706 | −3.550* | |||||||
| Distance (km) from contaminated seashore (ref. > 0.80) | |||||||||
| ≤ 0.20 | −0.631 | −6.412* | −1.629* | ||||||
| 0.21–0.40 | 0.121 | 0.498 | −1.819 | ||||||
| 0.41–0.60 | −0.532* | 2.040 | −0.671 | ||||||
| 0.61–0.80 | −0.799* | −1.248 | 0.201 | ||||||
| Presence of respiratory symptoms | 0.095* | 0.704 | |||||||
| Risk of PTSD | 0.740 | ||||||||
| R2 | 0.018 | 0.025 | 0.020 | 0.053 | 0.021 | 0.071 | 0.006 | 0.044 | 0.215 |
| F statistics | 3.028* | 4.426* | 3.549 | 4.077* | 4.804* | 2.984* | 3.078* | 6.372* | 13.853* |
Figure & Data
References
Citations

- Single and mixed effects of multiple volatile organic compounds exposure on hematological parameters in the U.S. adult population
Yan Zhuang, Laifu Li, Yanqi Zhang, Xuna Liu, Beibei Zeng, Boxu Zhu, Fei Dai
Chemosphere.2024; 355: 141825. CrossRef - Latent Trajectories of Haematological, Hepatic, and Renal Profiles after Oil Spill Exposure: A Longitudinal Analysis
Benjamin Atta Owusu, Apiradee Lim, Nitinun Pongsiri, Chanthip Intawong, Sunthorn Rheanpumikankit, Saijit Suksri, Thammasin Ingviya
International Journal of Environmental Research an.2023; 20(4): 2871. CrossRef - Haematological, renal, and hepatic function changes among Rayong oil spill clean-up workers: a longitudinal study
Benjamin Atta Owusu, Apiradee Lim, Chanthip Intawong, Sunthorn Rheanpumikankit, Saijit Suksri, Thammasin Ingviya
International Archives of Occupational and Environ.2022; 95(7): 1481. CrossRef - Association between BTEX (benzene, toluene, ethylbenzene and xylene) concentration in ambient air with hematological and spirometric indices: a population-based study
Hosna Moradkhani, Mostafa Leili, Jalal Puralajal, Ashraf Mazaheri Tehrani, Ayat Hossein Panahi, Mohammd Taghi Samadi, Sara Beheshtifar
Human and Ecological Risk Assessment: An Internati.2022; 28(5-6): 490. CrossRef - Combine effect of exposure to petrol, kerosene and diesel fumes: On hepatic oxidative stress and haematological function in rats
Solomon E Owumi, Bidemi N Oladimeji, Tobiloba C Elebiyo, Uche O Arunsi
Toxicology and Industrial Health.2021; 37(6): 336. CrossRef - Associations between blood volatile organic compounds, and changes in hematologic and biochemical profiles, in a population-based study
Sabit Cakmak, Christie Cole, Chris Hebbern, Julie Andrade, Robert Dales
Environment International.2020; 145: 106121. CrossRef - Developing Large-Scale Research in Response to an Oil Spill Disaster: a Case Study
Richard K. Kwok, Aubrey K. Miller, Kaitlyn B. Gam, Matthew D. Curry, Steven K. Ramsey, Aaron Blair, Lawrence S. Engel, Dale P. Sandler
Current Environmental Health Reports.2019; 6(3): 174. CrossRef


 PubReader
PubReader Cite
Cite
