JYNNEOS vaccine safety monitoring in the Republic of Korea, 2022: a cross-sectional study
Article information
Abstract
Objectives
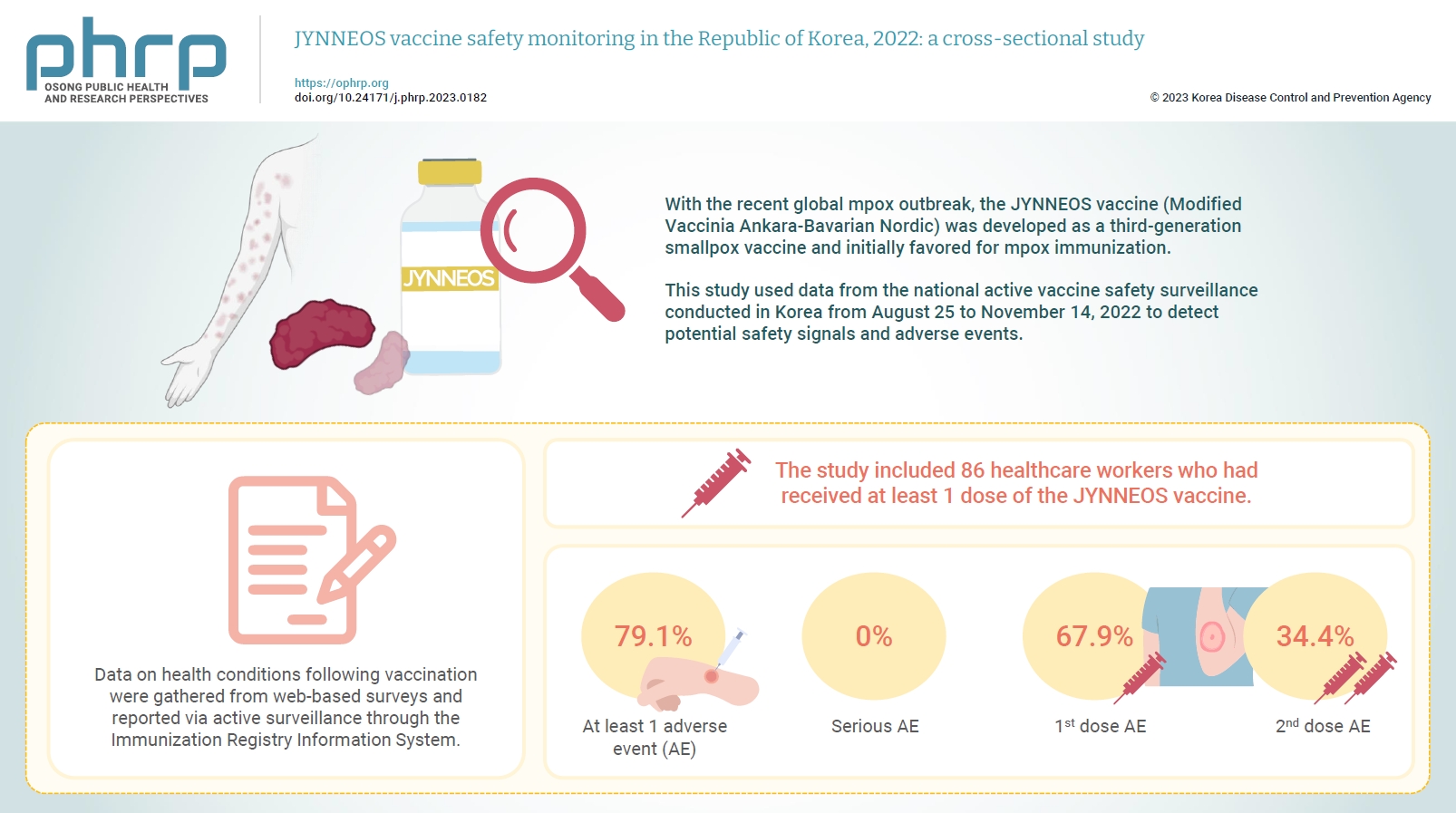
With the recent global mpox outbreak, the JYNNEOS vaccine (Modified Vaccinia Ankara-Bavarian Nordic) was developed as a third-generation smallpox vaccine and initially favored for mpox immunization. Vaccine-associated side effects contribute to vaccine hesitancy. Consequently, tracking adverse events post-immunization is crucial for safety management. This study used data from the national active vaccine safety surveillance conducted in Korea from August 25 to November 24, 2022 to detect potential safety signals and adverse events.
Methods
Data on health conditions following vaccination were gathered from web-based surveys and reported via active surveillance through the Immunization Registry Information System. This follow-up system functioned via a text message link, surveying adverse events and health conditions beginning on the second day post-vaccination. Information about specific adverse events, including both local and systemic reactions, was collected.
Results
The study included 86 healthcare workers who had received at least 1 dose of the JYNNEOS vaccine. Among the respondents, 79.1% reported experiencing at least 1 adverse event, with the majority being local reactions at the injection site. The incidence of adverse events was higher following the first dose (67.9%) than after the second dose (34.4%). The most frequently reported adverse event for both doses was mild pain at the injection site.
Conclusion
The study provides crucial information on the safety of the JYNNEOS vaccine, demonstrating that most adverse events were manageable and predominantly localized to the injection site. Nonetheless, additional research is needed on the safety of various vaccine administration techniques and the vaccine's effects on broader demographics.
Introduction
The first case of mpox in Korea was confirmed on June 22, 2022. In response, the Korea Disease Control and Prevention Agency (KDCA) sought emergency use authorization for JYNNEOS, a third-generation smallpox vaccine derived from a live, attenuated orthopoxvirus, known as modified vaccine Ankara. By July 1, 2022, the Ministry of Food and Drug Safety (MFDS) had approved the emergency use of JYNNEOS, following a review by the Korea Orphan and Essential Drug Center. The vaccine was administered to adults aged ≥18 years as a 2-dose series (0.5 mL per dose) via subcutaneous injection, with doses given at least 28 days apart. The KDCA guidelines also specify that individuals who have previously been vaccinated against smallpox should receive only 1 dose [1]. Given the potential for exposure among laboratory and healthcare personnel due to their occupational activities, many countries recommend prioritizing these workers for vaccine access [2-4]. Accordingly, the KDCA’s mpox vaccination strategy primarily targets individuals who have known or presumed exposure to mpox, or those who are at high risk for occupational exposure, such as healthcare workers, laboratory workers, and Epidemiological Investigation Officers [1-5]. This report presents an analysis of adverse event data collected from healthcare workers who received the JYNNEOS vaccination between August 25 and November 24, 2022.
Materials and Methods
Data Collection
Between August 25 and November 24, 2022, a total of 99 individuals, specifically those at occupational risk of exposure, voluntarily received at least 1 dose of the JYNNEOS vaccine at a healthcare center in Korea. For the analysis, we included 86 individuals who reported their health conditions after receiving at least 1 dose. The KDCA actively monitored the safety of the JYNNEOS vaccine. The day after their vaccination, individuals received text message notifications containing a health survey, which asked about potential adverse events for the 7 days following vaccination. This questionnaire was based on the Korean MFDS’s guideline for evaluating the severity of adverse events in vaccine clinical trials, and was used to evaluate serious adverse events [2]. The survey assessed the severity of 12 symptoms and signs. In each survey, participants were asked if they were experiencing symptoms, and the severity of each symptom was classified as either mild, moderate (symptoms that do not interfere with daily life), or severe (symptoms that interfere with daily activities and necessitate emergency visits). During the surveillance period, all health conditions were recorded, even for those without symptoms.
Study Design
This study was carried out as a prospective, self-reported survey of adverse events over a period of 121 days, from August 2022 to November 2022. The data reported were analyzed according to age group (20−29, 30−39, 40−49, 50 years and above), dosage, severity of adverse events, and symptoms indicative of potential adverse events. Descriptive statistics were utilized to detail the adverse events experienced by the participants. All analyses were performed using SAS ver. 9.4 (SAS Institute).
IRB Approval
All activities described in this study were carried out under the supervision and approval of the public health authority. In line with government regulations, informed consent was duly obtained. The study, which involved text message-based surveillance, was granted an exemption from review by the Institutional Review Board of the KDCA (2022-08-09-PE-A).
Results
After applying the exclusion criteria detailed in Figure 1, we included a total of 86 individuals who reported their health conditions following at least 1 dose of the vaccine. Of these 86 respondents, 70 (81.4%) were fully vaccinated with a second dose, while 16 (18.6%) were partially vaccinated with just 1 dose due to the presence of a smallpox vaccination scar, which is a clear indicator of previous vaccination. All of the vaccine recipients were healthcare workers, including Epidemiological Investigation Officers. Among them, 51.2% were women, and 38.4% were aged between 30 and 39 years. A summary of the characteristics of the vaccine recipients is presented in Table 1.
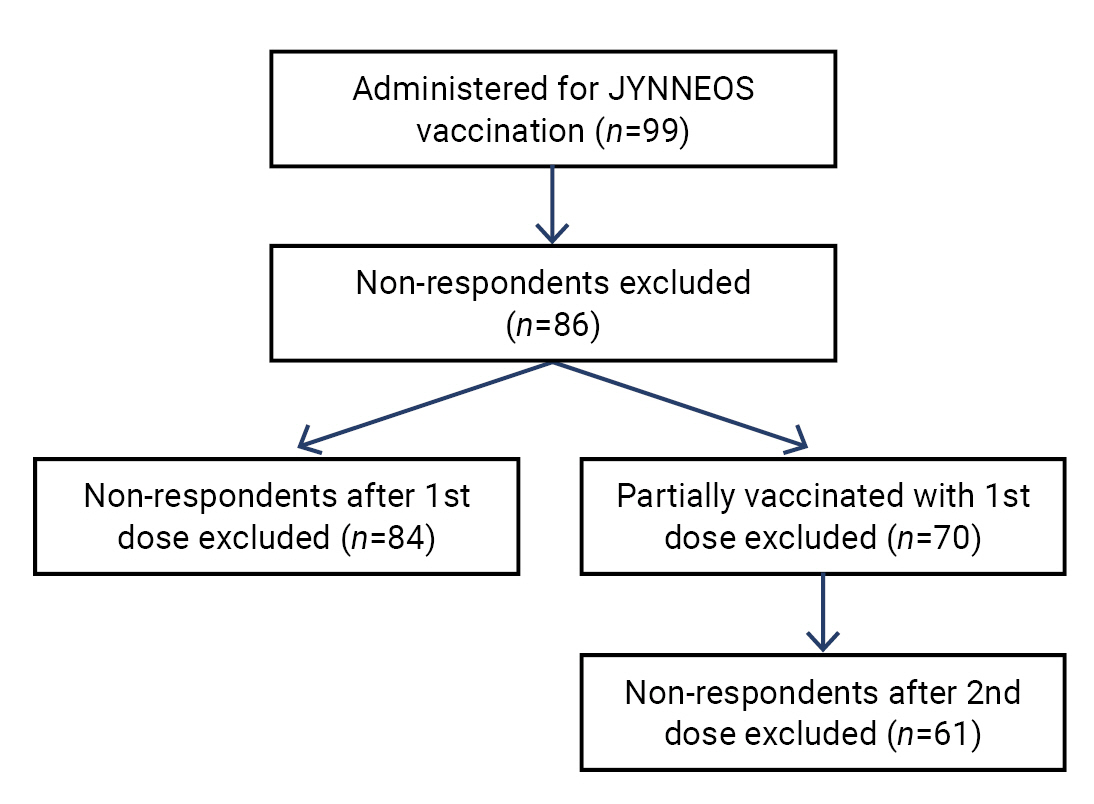
Flowchart illustrating the exclusion of sample subjects from the pool of vaccinated recipients.
Between August 25 and November 24, 2022, JYNNEOS vaccinations were administered to 99 individuals. Thirteen participants who did not report any consultations for any of the listed adverse events were excluded. Additionally, we had to exclude those with missing reports for each dose. After excluding non-respondents (n=2), the analysis of the first dose included 84 individuals. For the analysis of the second dose, we excluded those who had not received the second dose (n=16) and non-respondents (n=9), leaving 61 individuals included in the final analysis.

Characteristics of JYNNEOS vaccine recipients with reports submitted to Immunization Registry Information System after vaccination (n=86), Republic of Korea, August 25 to November 24, 2022
Adverse Events by Signs and Symptoms
Of all the reports, 79.1% documented at least 1 adverse event (Table 1). The most commonly reported adverse events were local reactions, including pain, swelling, and erythema at the injection site. Systemic reactions such as pyrexia, fatigue, headache, and myalgia were reported infrequently. Regarding gastrointestinal disorders, there were no reports of nausea, but sporadic cases of diarrhea were noted, ranging from a minimum of 1 case (1.6%) to a maximum of 2 cases (2.4%). Skin disorders (excluding those at the injection site) were very rare, with a minimum of 1 case (1.6%) and a maximum of 7 cases (8.3%). These symptoms were mild and did not interfere with the individuals’ daily activities. There were no reports of severe neurological disorders; however, mild symptoms, such as sweating and dizziness, were reported, ranging from a minimum of 1 case (1.6%) to a maximum of 2 cases (2.4%) (Table 2).
Incidence of Adverse Events between First and Second Vaccine Doses
The incidence of adverse events following the first dose was 67.9%, and 34.4% after the second dose. The risk seems to be relatively lower after the administration of the second dose (Table 3). The most commonly reported adverse events for both the first and second doses were similar. The majority of these adverse events were local reactions, with the most common being pain, swelling, and erythema at the injection site. Following the first dose, the incidence of pain at the injection site was over 64.3%. Most of these were instances of mild pain that did not interfere with daily activities (n=51, 60.7%), and very few were reported as pain that interfered with daily activities (n=4, 4.8%). For the second dose, nearly half (49.2%) reported mild pain at the injection site that did not interfere with daily life. The incidence of swelling and erythema at the injection site after the first and second doses were relatively similar. However, the size of the affected areas with swelling and erythema appeared to be larger after the second dose. No serious adverse events were reported following either the first or second vaccinations. Table 2 presents the reporting rates for the first and second doses by symptom.
Discussion
We conducted the first study aimed at identifying adverse events in healthcare workers in Korea who had received a minimum of 1 dose of the JYNNEOS vaccine. The web-based survey was dispatched the day following their vaccination, and it required reporting for at least 1 day after each dose. The majority of adverse events were reactions at the injection site, and no new worrisome symptoms were identified. This aligns with the findings of previous studies [7,8].
This study offers timely insights into the safety of mpox vaccination. However, it should be noted that the findings detailed in this report are subject to several limitations, which are addressed below alongside recommendations for best practices in future research. First and foremost, only reactions at the subcutaneous injection site were observed. In a previous study, vaccine administration errors were reported more frequently following intradermal administration than subcutaneous administration of the JYNNEOS vaccine [7]. Given that the mpox vaccination was administered solely subcutaneously during the study period, data on adverse events following alternative routes of administration is limited. Further research is necessary to examine the effects of different injection techniques. This could contribute to a more comprehensive understanding of the safety of the method, particularly in comparison to intradermal injection.
Secondly, the study population was composed exclusively of healthy adults. Throughout the study period, mpox vaccinations were restricted and recommended solely for individuals with a high likelihood of exposure, such as those with occupational risks. The recipients of the vaccine were either healthcare workers, laboratory workers, or epidemiological investigation officers. As a result, there was a lack of diversity among the research participants. Concerns about vaccine safety could also hinder vaccination efforts; further studies are suggested to ensure the safety of vaccination for vulnerable populations, such as those with HIV or within specific age groups [9].
The safety data from the KDCA for the JYNNEOS vaccine indicate that the subcutaneous administration of the JYNNEOS mpox vaccine is safe. It further recommends the use of the JYNNEOS vaccine in accordance with KDCA guidelines. The KDCA will persist in monitoring any adverse events to provide vaccination recommendations that prioritize safety.
HIGHLIGHTS
• The safety of the JYNNEOS vaccine in response to an mpox outbreak was investigated in Korea from August to November 2022.
• Post-vaccination health conditions were monitored utilizing web-based surveys and the Immunization Registry Information System.
• Among the 86 healthcare workers included in the study, 79.1% reported experiencing adverse events, primarily localized at the injection site, and a higher incidence was observed after the first dose.
• Most of these events were manageable, but further research on diverse populations and administration methods is needed.
Notes
Ethics Approval
All activities described in this study were reviewed and conducted with authorization from the public health authority. Informed consent was collected in accordance with government regulations, and the study with the text message-based surveillance was exempted from review by the Institutional Review Board of KDCA (2022-08-09-PE-A).
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding
None.
Availability of Data
The data used in this study are protected under the Personal Information Protection Act.
Authors’ Contributions
Conceptualization: JL, JP, HB, GYK; Data curation: JL, JP, HB; Formal analysis: JL, JP, HB; Investigation: JL, JP, HB; Methodology: all authors; Supervision: GYK; Validation: JL, JP, HB, GYK; Visualization: JL; Writing–original draft: JL; Writing–review & editing: all authors. All authors read and approved the final manuscript.