Effectiveness of virtual reality-supported exercise therapy in improving upper extremity function and activities of daily living among patients after stroke: a systematic review of randomized control trials
Article information
Abstract
This systematic review describes the effectiveness of virtual reality (VR)-supported exercise therapy on upper limb motor function and activities of daily living after stroke. Studies published through January 24, 2022, were identified using CINAHL, Cochrane Library, Embase, Medline, and Web of Science. Randomized control trials comparing VR treatment with conventional therapy (CT) for upper extremity rehabilitation after stroke were included. Methodological quality was assessed using the Cochrane risk-of-bias tool. Of 9 included studies, 5 concluded that the VR group outperformed control participants, 1 indicated the superiority of VR-supported exercises alone over CT, and 3 found VR comparable to CT in promoting upper limb motor function. Five studies analyzed independence in daily living, with 4 reporting no significant difference between VR and CT groups. No strong evidence indicated long-term benefits of VR-assisted exercise. All included studies demonstrated low risk of bias concerning random sequence generation, allocation concealment, outcome assessment blinding, incomplete outcome data, and selective reporting bias. However, a high risk of bias was observed regarding participant blinding due to the nature of the intervention. Most studies suggested that VR, used alongside CT, can improve motor function following stroke. However, the evidence was insufficient to conclude that VR outperforms conventional approaches.
Introduction
Stroke is the second leading cause of death worldwide, with an annual mortality figure of approximately 5.5 million deaths [1]. Additionally, up to 50% of survivors experience a permanent disability [2]. Stroke is characterized by neurological deficits and long-term disability resulting from a focal injury to the central nervous system, such as cerebral infarction, intracerebral hemorrhage, or subarachnoid hemorrhage [3].
Changes related to impaired upper arm function include difficulty performing tasks, abnormal postural adjustments, an inability to control grasping, and functional disability. These challenges make it difficult for individuals to participate in activities of daily living (ADLs) such as eating, dressing, and grooming [4,5]. Research indicates that 40% of stroke survivors experience reduced function in the affected arm, and approximately 70% of patients report diminished arm function [6]. Separately, over 80% of patients have been found to exhibit impaired upper limb motor function after stroke, with 66% continuing to have a non-functional upper limb long-term [7,8]. Upper limb impairments are often more pronounced than those of the lower limb [9] and include functional limitations and uncoordinated motion [10]. Conventional rehabilitation techniques such as physiotherapy, neurodevelopmental therapy, and proprioceptive neuromuscular facilitation are effective to similar degrees in improving motor function. However, these methods can be resource-intensive and costly, often requiring specialized facilities that are not widely available. Virtual reality (VR) has emerged as a novel rehabilitation strategy and an enjoyable alternative to enhance motor recovery after stroke [11,12]. VR is an emerging technology that provides an interactive simulation, creating an environment that closely mimics reality for users. VR has been described as “the use of interactive simulations created with computer hardware and software to present users with opportunities to engage in environments that appear and feel similar to real-world objects and events” [13]. VR is based on several principles, including goal-oriented tasks, adjustable task difficulty, task repetition, feedback, and the promotion of increased user motivation and enjoyment [14]. VR systems can be categorized as immersive or non-immersive. Non-immersive VR systems allow users to interact with the virtual environment displayed on a computer screen while maintaining a sense of the real world. Immersive VR systems, which include large screen projections, head-mounted displays, or Cave Automatic Virtual Environment (Balance Near Automatic Virtual Environment) systems, block out all real-world perception, immersing the user solely in computer-generated imagery [15].
Adults with persistent hemiparesis have recently been shown to benefit from VR therapy, with reasonably good patient compliance [16]. Engaging in physical exercises through VR programs can help patients with neurological disorders enhance their motor learning and control, as well as their functional capacities [17]. Research suggests that training in fine hand-motion rehabilitation can be more effective when it incorporates immersive VR technology [18]. Several studies have indicated that VR technology produces better outcomes than conventional therapies in improving upper extremity function [19–24] and ADL performance [22–24] after stroke. However, a systematic review conducted by Laver et al. [25] reported no appreciable difference between VR-based training and conventional treatment in the recovery of upper limb motor function. Due to the variability of findings regarding VR intervention, a systematic review is necessary to thoroughly explore the effects of VR rehabilitation on upper arm function. The aim of this review was to evaluate the benefits of VR in the motor recovery of upper limb function and in promoting independence in ADLs, focusing on studies conducted a maximum of 2 years after stroke.
Materials and Methods
Eligibility Criteria
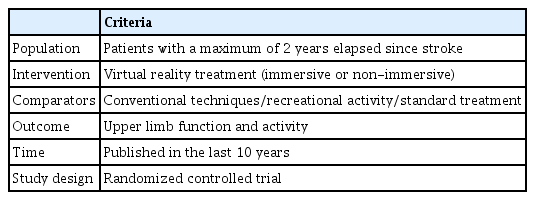
We conducted this systematic review in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (Supplementary Material 1). We incorporated studies that met the following inclusion criteria: (1) randomized controlled trials (RCTs) that compared VR with conventional interventions in upper limb rehabilitation, with no more than 2 years elapsed since the stroke, and that met predefined PICOTS criteria (Table 1); (2) the intervention group received VR-supported exercise therapy either alone or in combination with conventional therapy, while the control group received conventional therapy, recreational therapy, or standard care alone; (3) the study participants were at least 18 years old with any type of stroke (hemorrhagic or ischemic); and (4) the article was accessible and written in English. We excluded studies that compared 2 types of VR treatment, those involving robotics-supported therapy or VR treatment administered through telerehabilitation, and those conducted in home settings. Studies that included participants with cognitive impairments and those that did not focus exclusively on upper limb function were also excluded.
Search Strategy
This review was conducted between October 2022 and March 2023. All reviewers independently performed a literature search across 5 databases: CINAHL, Cochrane Library, Embase, Medline, and Web of Science. The keywords used for the literature search were “stroke,” “cerebrovascular accident,” “virtual reality,” “virtual environment,” “motor deficits,” “neurorehabilitation,” and “upper limb function.” These keywords were used either individually or in combination, employing the Boolean operators “OR” and “AND”. No restrictions were imposed on the year of publication, and the most recent study publication date was January 24, 2022.
Outcome Measures
The primary outcome was upper limb function and activity, while the secondary outcomes were independence in daily living and the long-term benefits of VR therapy. We conducted a narrative synthesis of data from the identified studies, which encompassed participant characteristics, attributes of the interventional and control groups, upper limb function and activity, independence in ADLs, and the effects of VR therapy during the follow-up period.
Data Collection and Processing
The screening procedure was performed independently by 2 reviewers to identify relevant studies based on predetermined eligibility criteria. Initially, the titles of potential studies were screened, followed by an examination of the abstracts and full texts of the articles for inclusion in the review. Studies were assessed against the inclusion and exclusion criteria before being selected. Data from the articles were extracted and analyzed independently by the first 2 reviewers, with their findings subsequently verified by a third reviewer. Each reviewer examined the data individually to lead to a consensus while minimizing bias. After extracting data from all included articles, a table was created. This table included the author, publication year, country of origin, number of participants, type of VR used, nature and duration of the intervention, outcome variables and scales used, and results for each study (Table 2) [11,26–33].
Assessment of Study Risk of Bias
The risk of bias in the selected documents was assessed using the Cochrane risk-of-bias tool [34]. The evaluation covered several areas: allocation concealment and random sequence generation (both related to selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), completeness of outcome data (attrition bias), and selective data reporting (reporting bias) (Table 3) [11,26–33]. The level of bias was categorized into 3 grades: low, high, and unclear risk of bias. The reviewers independently assessed the biases in the included studies and, in the event of any discrepancies, consulted a third reviewer for resolution.
Data Synthesis
We conducted a narrative synthesis of data from the selected studies, encompassing participant characteristics, descriptions of the intervention and control groups, details of the VR interventions, and comparisons between VR and conventional therapy. This synthesis also included assessments of upper limb function and activity, independence in ADLs, and the impact of VR therapy over the course of follow-up.
Results
A total of 589 relevant studies were identified. Of these, 412 were duplicates, 30 did not meet the eligibility criteria, and 112 were excluded after screening titles and abstracts. Ultimately, 35 studies were assessed for eligibility, and 9 RCTs were included in the systematic review.
Study Characteristics
The studies included in this review were conducted in 7 countries: 2 studies in the Republic of Korea [26,27], 2 in China [28,29], 1 in Canada, 1 in Norway [30], 1 in Saudi Arabia [31], 1 in the United Kingdom [32], and 1 in Turkey [33]. The total sample size across the included articles was 558 participants, with 282 in the experimental groups and 276 in the control groups. The time since stroke onset ranged from 9 days to 24 months. Six studies [11,26,28–30,33] recruited participants within 6 months after stroke, while 3 trials [27,31,32] included participants for whom more than 6 months had elapsed. In 1 study [32], the intervention and control groups were subdivided into patients with ischemic and hemorrhagic stroke, whereas the other studies did not report specific details about the type of stroke. The frequency of training varied from 3 to 5 sessions per week, and the number of intervention sessions ranged from 10 to 36. The duration of each session varied from 30 minutes to 120 minutes. In 1 study [30], the experimental and control groups were further segmented based on the severity of hand paresis, specifically into mild to moderate hand paresis and severe hand paresis categories. Few studies investigated whether the benefits of VR treatment were sustained in the long term after the therapy concluded. One study [30] reassessed outcomes 3 months post-intervention, while another study [11] conducted an assessment 1 month after the intervention ended.
Risk of Bias and Study Quality
The risk of bias for the included studies is detailed in Table 3 and Figure 1 [11,26–33]. All 9 studies employed random sequence generation, allocation concealment, and blinding of outcome assessors. None of the studies exhibited selective reporting or attrition bias, the latter of which pertains to participant dropout. In 7 studies [11,26,27,29,31-33], the participants and personnel were not blinded to the allocated intervention. For the remaining 2 studies [28,30], the risk of performance bias was unclear. Therefore, a high risk of performance bias was found overall, since blinding of participants was not feasible due to the nature of the intervention. Apart from performance bias, all included studies were assessed as having a low risk of bias in all other domains. The outcome assessors were adequately blinded by withholding knowledge of the allocated interventions. Based on the quality assessment of the included studies, the total bias score was determined to be 1 out of 7, indicating a low risk of bias.
Intervention Group Characteristics
In 7 studies [26–28,31–33], the intervention group received a combination of conventional treatment and VR therapy, while in 2 studies [29,30] the intervention group was treated exclusively with VR. The VR-based interventions employed in these studies utilized systems that provide feedback within a virtual environment, including You Grabber [30], Armeo Spring [31], VR-based bilateral upper extremity training (VRBT) [27], A2, YiKang, reinforced feedback in a virtual environment (RFVE) [32], a Leap Motion-based VR system [28], and the Interactive Rehabilitation and Exercise? system [26]. Other studies involved the intervention group using commercially available gaming systems for VR-based therapy, such as the Wii (Nintendo) [11] and the Xbox Kinect [33], within a VR environment.
Control Group Characteristics
In 5 studies [11,29–32], interventions were administered to both groups with matching frequency (number of sessions per week), session duration, and treatment duration. However, in the remaining 4 studies [26–28,33], the duration of therapy for the control group did not match that of the intervention group.
Measurement Tools
A variety of scales were used in the 9 studies to assess improvements in upper limb motor function, arm function, and ADLs. The scales employed included the Fugl-Meyer Assessment-Upper Extremity (FMA-UE) [26,29,32,33], the Box and Block Test (BBT) [11,27,30,33], the Functional Independence Measure (FIM) [11,30,32,33], the Barthel index [11], the Korean version of the modified Barthel index [26], the Action Research Arm Test (ARAT) [29–31], the Wolf Motor Function Test (WMFT) [11,28,31], hand grip strength [31], the Grooved Pegboard Test [27], the Jebsen–Taylor Hand Function Test [27], the Brunnstrom stage scale [33], the Stroke Impact Scale (SIS) [11], the Edmonton Symptom Assessment System (ESAS) [32], the Manual Function Test [26], and a dynamometer [11].
Comparison of VR Therapy versus Conventional Therapy/Standard Care/Recreational Therapy
The results of this comparison are detailed in the following sections.
Upper limb function and activity
Upper limb function and activity were assessed in all studies. Of the 9 articles, 5 [27,28,31–33] concluded that the stroke survivors receiving VR treatment experienced better outcomes than those administered conventional treatment. One study [29] reported that VR-supported exercises alone were superior to conventional therapy, while 3 studies [11,26,30] indicated that VR training was as effective as conventional methods.
A study by Ikbali Afsar et al. [33] reported better outcomes in the interventional group, with significant differences observed between the experimental and control groups in terms of FMA-UE and BBT scores (p<0.05). Similarly, Wang et al. [28] concluded that the motor function of the affected upper limbs, as measured with the WMFT scale, was significantly improved in the experimental group compared to the control group (p<0.01). Additionally, the WMFT time was significantly shorter in the experimental group (3.29±0.82 seconds) than in the control group (4.24±0.57 seconds) post-intervention. El-Kafy et al. [31] found comparable results, stating that patients in the experimental group showed more favorable improvement than those in the control group in terms of the ARAT (p<0.01), WMFT (p<0.01), and WMFT-Time (p<0.01) assessments. They concluded that the use of combined therapy (VR and conventional rehabilitation) provides greater benefits to upper limb function than conventional treatment alone. Kiper et al. [32] also reported that the experimental group demonstrated better improvement on the FMA-UE scale (p<0.001), FIM (p<0.001), National Institutes of Health Stroke Scale (p≤0.014), and ESAS (p≤0.022) compared to the control group. Kiper et al. [32] further assessed kinetic parameters such as time (p<0.001), speed (p<0.001), and peak (p<0.001) during the execution of the requested task, finding significant differences compared to control participants. Lee et al. [27] reported that members of the VRBT group exhibited significant improvements in upper extremity function and muscle strength (p<0.05). Lastly, Chen et al. [29] described a significant improvement in the ARAT (Z=2.088, p=0.04) and FMA-UE (Z=−2.338, p=0.02) scales in the VR group compared to the control group.
Three studies reported nonsignificant results [11,26,30]. Kwon et al. [26] observed improvements in both groups post-intervention, and the difference between them was not statistically significant (p>0.05). The effect size of the change in the examined variable was medium (Cohen d=0.48) for the FMA. Similarly, Saposnik et al. [11] found that the WMFT performance time improved from baseline to post-intervention for both groups. Specifically, they observed a decrease in the median time from 43.7 seconds (interquartile range [IQR], 26.1–68.0 seconds) to 29.7 seconds (IQR, 21.4–45.2 seconds), representing a 32.0% reduction, for the VRWii group, compared to a change from 38.0 seconds (IQR, 28.0–64.1 seconds) to 27.1 seconds (IQR, 21.2–45.5 seconds), representing a 28.7% reduction, for the patients treated with recreational activity. However, they noted no significant difference in hand function (p=0.314), grip strength (p=0.713), or motor performance (p=0.469) between the groups at the end of the intervention, except for gross manual hand dexterity as measured by the BBT scale (p=0.018). Brunner et al. [30] reported that participants in the VR group improved on the ARAT by 12 points (standard deviation [SD], 11) from baseline to the post-intervention assessment, while the patients receiving conventional therapy improved by 13 points (SD, 10), with no significant difference (p=0.714) between the control and experimental groups.
Independence in daily living
Of the included studies, 5 [11,26,30,32,33] analyzed the impact of VR therapy versus conventional therapy on ADLs. Four of these studies [11,26,30,33] reported no significant differences in ADL performance between the control and experimental groups. Supporting this, Ikbali et al. [33] found no statistical difference (p=0.677) between the groups with regard to FIM score, although both the experimental (12.74±2.51 to 23.74±4.42) and control (13.63±3.61 to 23.63±4.99) groups showed improvement from baseline to post-intervention. Kwon et al. [26] also reported no significant difference in ADL between the VR and conventional therapy groups (p>0.05), despite a large effect size (Cohen d=1.02). Saposnik et al. [11] observed similar improvements in the Barthel index in the experimental (64.7±22.4 to 83.4±18.0) and control (64.2±23.0 to 80.3±21.7) groups. Brunner et al. [30] noted comparable improvements in FIM motor scores in the VR group (changing from 15.9 at baseline to 12.7 after intervention) and the conventional therapy group (changing from 17.5 at baseline to 13.1 after intervention) post-intervention and at follow-up. However, Kiper et al. [32] reported a significant improvement in independence in daily living associated with VR therapy, as measured with FIM (RFVE, 8.42%; conventional rehabilitation, 4.82%; p<0.001).
Upper limb function: follow-up
Only 2 studies [11,30] analyzed the long-term benefits of VR therapy among patients treated after stroke. Brunner et al. [30] observed an improvement in upper extremity function as assessed with ARAT, with a mean increase of 17 points (SD, 13) from baseline to follow-up in the VR group, which was identical to the improvement seen in the conventional therapy group. Consequently, no significant difference was found (p=0.777) between groups at follow-up when assessing all scales (ARAT, BBT, FIM, and Patient Global Impression of Change). Similarly, Saposnik et al. [11] found no significant differences at the 4-week post-intervention mark in WMFT performance time (p=0.346), Barthel index (p=0.774), FIM (p=0.848), or SIS hand function (p=0.514) between the control group (who engaged in recreational activity) and the experimental group (who received VRWii therapy).
Discussion
More than three-quarters of stroke victims experience diminished upper limb function following the event, impacting their everyday quality of life. The aim of this study was to compare motor recovery of upper limb function between interventional (VR therapy) and control (conventional therapy) groups. The interventional group received both conventional treatment and VR therapy in 7 studies [26–28,31–33], while only VR therapy was administered in 2 studies [29,30].
Of the 9 studies examined, 5 [27,28,31–33] concluded that the VR treatment group of stroke survivors experienced better outcomes than those who received conventional treatment. One study [29] reported the superiority of VR-supported exercises, while 3 studies [11,26,30] found that VR training was similarly effective to conventional methods. Most of the included studies [27–29,31–33] noted that the intervention group experienced greater improvements in upper limb function and activity than the control group. According to El-Kafy et al. [31], interventional groups using combination therapy (both virtual and traditional) saw greater enhancements in upper limb function. All trials considered reported a similar degree of improvement in ADLs between groups. Vinas-Diz and Sobrido-Prieto [35] have emphasized that VR can create environments specifically tailored for patients by modifying and controlling their engagement with their surroundings, potentially serving as a valuable adjunct or alternative to conventional therapy. In a separate comprehensive review, Zhu et al. [36] found that VR intervention is a beneficial non-pharmacological approach for enhancing cognitive and motor performance in older individuals. Wiley et al. [37] observed that the VR group performed better than the control when VR was used as an add-on to conventional therapy, concluding that VR therapy is stimulating and interesting relative to traditional rehabilitation methods. These findings aligned with those of several included studies [27,28,31–33]. Zhang et al. [38] also reported that the upper limb function in the VR group outperformed that of the conventional method, with significant differences found between the 2 groups regardless of the intervention period. This was consistent with results from other included studies [27,28,31–33], which had varied intervention durations. Separately, Zhu et al. [36] concluded that VR treatment is beneficial for improving outcomes in patients with mild cognitive impairment after stroke. A systematic review by Gao et al. [39] concluded that VR-based training did not have significant benefits over conventional methods on motor function or ADLs. However, they suggested that greater duration, frequency, and daily intensity of VR training may yield better outcomes. Chen et al. [29] reported that although the VR group showed significant improvement in upper extremity motor function and independence in day-to-day activities, the results were inconclusive regarding the benefits of VR therapy after the intervention ended. This was consistent with findings from other included studies [11,30]. The strengths of this systematic review include the exclusive inclusion of RCTs and a rigorous quality assessment of included studies using the Cochrane risk-of-bias tool. This review highlights several opportunities for future research to evaluate the long-term benefits of VR-supported exercises. However, several limitations should be addressed. Most of the included studies had small sample sizes, which limited generalizability and weakened the validity of the results. Secondly, the intervention type (immersive or non-immersive VR), duration, and frequency varied between studies, potentially influencing rehabilitation progress. The optimal frequency and duration of intervention remain to be explored.
Implications for Practice
VR-based interventions can be incorporated into clinical practice to rehabilitate the upper limb in conjunction with conventional exercises. Commercially available games, such as Nintendo Wii Sports, are readily accessible and affordable, making them a potentially appealing choice. Specialized VR programs can also be tailored to the patient’s condition, enabling clinicians to effectively integrate VR treatment for rehabilitation after stroke.
Recommendations
The optimal frequency and duration of intervention using VR have yet to be determined, necessitating further research to compare the impacts of various VR interventions, including their respective frequencies and durations. Moreover, a need exists for studies with larger sample sizes to evaluate the long-term effects of VR-supported exercises. Additionally, the suitability of these interventions following stroke warrants further investigation to ascertain which patient groups are most likely to benefit and to determine the optimal timing for initiating intervention across the stages of stroke recovery (acute, subacute, and chronic). Research should also be conducted to examine the potential adverse effects associated with VR-supported exercises.
Conclusion
Most studies included in this review suggest that incorporating VR-supported exercise alongside conventional therapy may enhance the improvement of upper limb function and activity. However, the long-term benefits of VR-supported exercises among patients after stroke remain inconclusive.
HIGHLIGHTS
• Healthcare providers can integrate virtual reality (VR)-supported exercises into rehabilitation programs for survivors of stroke.
• The use of VR as a supplement to conventional therapy shows promise as an adjuvant for this patient population.
Supplementary Material
Supplementary data are available at https://doi.org/10.24171/j.phrp.2023.0148.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 checklist.
Notes
Ethics Approval
Not applicable.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding
None.
Availability of Data
The datasets used in this study are not publicly accessible; however, they can be obtained from the corresponding author upon reasonable request.
Authors’ Contributions
Conceptualization: PD, UP; Data curation: PD, SPS; Formal analysis: PD, NK; Investigation: UP; Methodology: PD, SPS; Project administration: NK, PD; Software: PD, NK; Supervision: NK; Validation: UP; Visualization: PD; Writing–original draft: PD, UP, NK; Writing–review & editing: all authors. All authors read and approved the final manuscript.