Quality of life in patients treated for COVID-19–associated mucormycosis at a tertiary care hospital
Article information
Abstract
Objectives
Coronavirus disease 2019 (COVID-19)–associated mucormycosis (CAM) has emerged as a formidable infection in patients with COVID-19. The aggressive management of CAM affects quality of life (QOL); thus, this study was designed to assess the QOL in patients with CAM at a tertiary healthcare institution.
Methods
This cross-sectional study of 57 patients with CAM was conducted over 6 months using a semi-structured standard questionnaire (the abbreviated World Health Organization Quality of Life questionnaire [WHO-BREF]) and a self-rated improvement (SRI) scale ranging from 0 to 9. Cut-off values of ≤52 and <7 were considered to indicate poor QOL and poor improvement, respectively. The correlations of QOL and SRI scores were evaluated using Spearman rho values.
Results
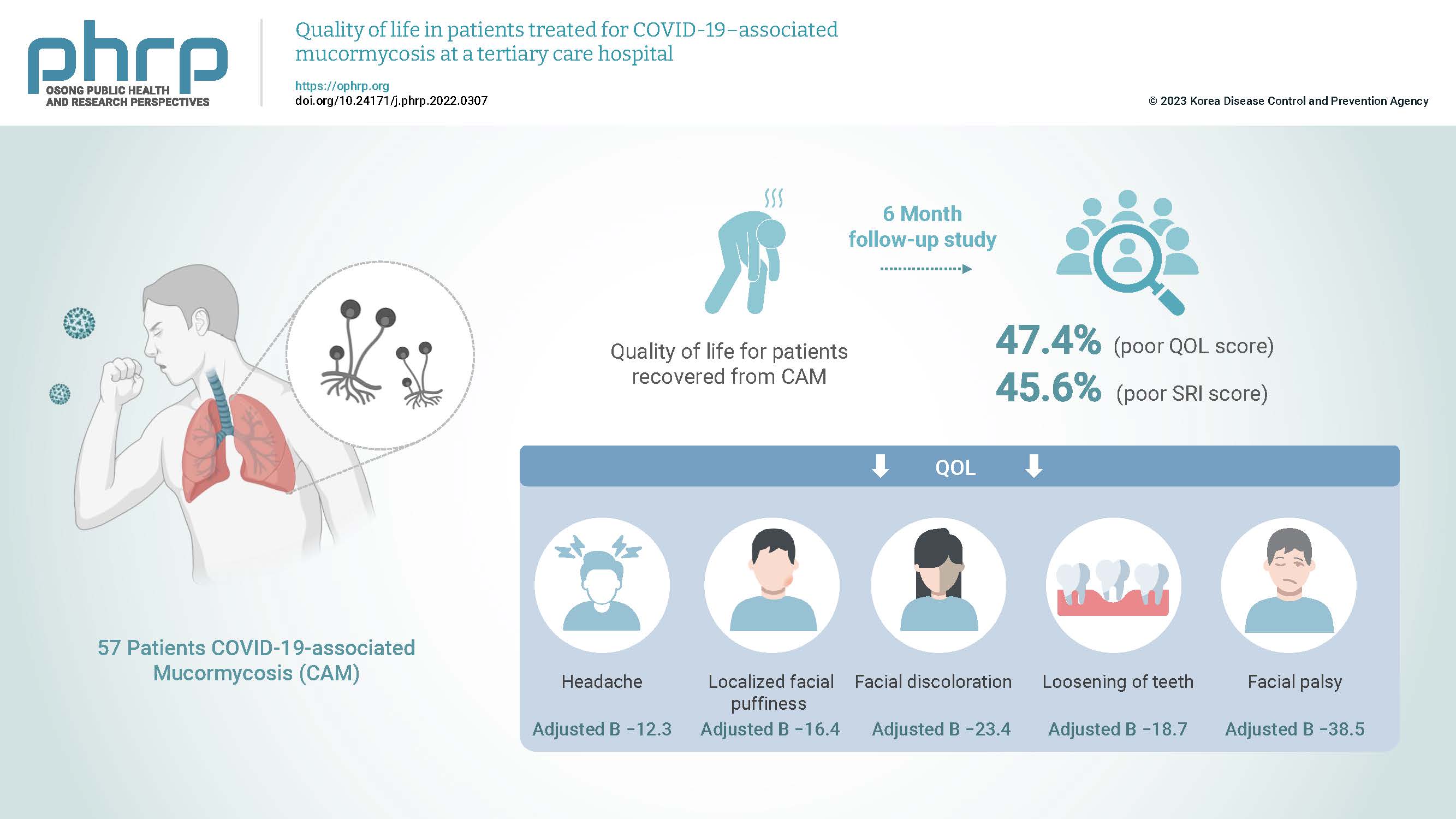
In total, 27 patients (47.4%; 95% confidence interval [CI], 34.9%–60.1%) and 26 patients (45.6%; 95% CI, 33.4%–58.4%) had poor QOL and poor SRI scores, respectively. The overall median (interquartile range) QOL score was 52 (41–63). Headache (adjusted B, −12.3), localized facial puffiness (adjusted B, −16.4), facial discoloration (adjusted B, −23.4), loosening of teeth (adjusted B, −18.7), and facial palsy (adjusted B, −38.5) were significantly associated with the QOL score in patients with CAM.
Conclusion
Approximately 1 in 2 patients with CAM had poor QOL and poor improvement. Various CAM symptoms were associated with QOL in these patients. Early recognition is the key to optimal treatment, improved outcomes, and improved QOL in patients with CAM.
Introduction
During the coronavirus disease 2019 (COVID-19) pandemic, many fungal infections such as aspergillosis, invasive candidiasis, and mucormycosis have been reported, especially among patients with severe COVID-19 and those recovering from the disease [1]. Mucormycosis, also known as black fungus, is a rare angioinvasive fungal infection caused by a group of fungi termed mucoromycetes [1,2]. The incidence of mucormycosis has increased so dramatically that it has become a notifiable disease, increasing the overall disease burden. COVID-19–associated mucormycosis (CAM) has been reported in many countries, including Austria, Brazil, Egypt, France, India, Iran, Italy, and the United States [3,4]. A systematic review indicated that CAM constitutes 0.3% of COVID-19 coinfections [5]. The prevalence of mucormycosis in India has been estimated at approximately 140 per 1,000,000 population, which is nearly 80 times greater than the rate in developed countries [2,6]. Humans acquire the infection mainly via inhalation, ingestion, and traumatic inoculation, and generally only susceptible populations are infected.
India is one of the countries that have been most heavily impacted by COVID-19, with multiple waves of COVID-19 resulting in more than 100,000 deaths following the second wave in March 2021 [7,8]. This has been accompanied by an unanticipated increase in CAM cases. As a result, the Indian government has classified CAM as a notifiable illness, and numerous state governments have classified it as an epidemic [9].
Mucormycosis is not a new disease, and it primarily affects immunocompromised patients. With the second wave of COVID-19, the incidence of mucormycosis has increased following the injudicious use of steroids and monoclonal antibodies [10,11]. Leukopenia has also been observed in patients with COVID-19; this ultimately lowers the immune response, affecting multiple systems and increasing the risk of opportunistic infections including pulmonary (rhino-orbital-cerebral mucormycosis), gastrointestinal, integumentary, and disseminated diseases [12,13]. Globally, diabetes continues to be the primary risk factor for mucormycosis, which has a death rate of 46% [14]. The COVID-19 pandemic has increased the incidence of diabetes following the excessive use of steroids, further contributing to immune system impairment. The burden of diabetes in the Indian population is high, and diabetes increases people’s susceptibility to opportunistic infections, which disturb normal body parameters [15,16].
All of these factors increase the disease burden, leading to morbidity along with physical, social, and psychological consequences. Both the clinical manifestations of an invasive disease process and the treatments available for the management of mucormycosis (primarily surgical) can affect individuals’ psychological state; additionally, disability following extensive surgical procedures can impact social relations. These changes may impact the overall quality of life (QOL) [17]. CAM is associated with high mortality and morbidity, and its diagnosis is frequently missed in India [18].
The World Health Organization (WHO) defines health as not merely the absence of disease or infirmity, but rather a state of complete psychological, mental, and social well-being [19]. Patrick and Erickson [20] defined health-related QOL as the value assigned to the duration of life as modified by the impairments, functional states, perceptions, and social opportunities that are influenced by disease, injury, treatment, or policy. Most chronic illnesses can degrade overall health by impairing the ability to live comfortably, as well as limiting functional status, productivity, and QOL, and are significant drivers of medical expenses [21].
Several Indian studies [22–24] have assessed the clinical and epidemiological features of CAM, but none have examined the QOL of recovered patients. Thus, we planned to determine the overall QOL among patients with CAM at a 6-month follow-up and assess the improvement among these patients at a tertiary health care institution.
Materials and Methods
Study Design and Duration
This was a hospital-based cross-sectional study performed over 6 months (November 2021 to April 2022).
Study Setting
This study was conducted at the All India Institute of Medical Sciences (AIIMS) Patna, an institute of national importance under the Ministry of Health and Family Welfare Government of India. AIIMS Patna provides comprehensive health care to the people of Bihar. This institute was declared a dedicated COVID-19 hospital and a center of excellence for the treatment of CAM in Bihar [25]. A total of 200 confirmed cases of CAM were admitted through the Flu Clinic of the Department of Community and Family Medicine and were treated jointly by the departments of otorhinolaryngology, ophthalmology, neurology, and neurosurgery.
Study Participants
All patients with laboratory-/image-confirmed mucormycosis who were admitted, treated, and presented during the 6-month follow-up period at the otorhinolaryngology department were included in the study. Patients who did not consent to participate in the study and those who self-reported being under psychiatric care prior to infection were excluded from the study.
Sample Size and Sampling Technique
Previous studies reported diminished QOL in approximately 50% to 60% of patients who underwent oromaxillofacial interventions involving oral maxillofacial and eye surgery [26–29]. Thus, assuming that 55% of patients with CAM have compromised QOL, a minimum sample size of 71 would be required to reveal the outcome at a 95% confidence level, 20% relative precision, 20% refusal rate, and a population adjustment of 200, as determined using Statulator [30]. We included all patients with CAM who met the inclusion criteria and presented for follow-up at the otorhinolaryngology outpatient department (OPD) or the ophthalmology OPD of the institution during the study period. A designated clinic for follow-up with these patients was run by the otorhinolaryngology and ophthalmology OPD on 1 designated day per week (Wednesday). We used consecutive sampling to collect information from the patients.
Study Tools and Procedure
A predesigned, semi-structured, standard questionnaire was created to collect the details of the patients with CAM. The questionnaire consisted of multiple sections. Section A included sociodemographic details, such as the age, sex, occupation, education, and residential address of the patients. Section B included details of the clinical profile and course of hospital stay of the patients, such as COVID-19 vaccination status, comorbidity profile, persistent symptoms, disease severity based on symptoms (early, moderately advanced, or very advanced), mode of treatment (medical, surgical, or both), duration of hospital stay, and whether intensive care unit (ICU) admission was required. Section C included a self-rated improvement (SRI) score on a scale of 0 to 9, where 0 represents no improvement and 9 represents full improvement. Section D comprised questions about the QOL of the patients at 6-month follow-up using the WHOQOL BREF questionnaire [31]. The WHOQOL BREF tool contains 26 items on a 5-point Likert scale, and the score ranges from 0 to 100 after conversion. Higher scores represent better QOL. The scale measures physical, psychological, social, and environmental domains of QOL. The scale has been validated in an Indian setting with good internal consistency (0.86) [32].
The study tool was developed using Epicollect5 (Centre for Genomic Pathogen Surveillance) in the English language, and the CAM patients or the attendants accompanying them were interviewed face-to-face when they presented at the OPD for follow-up using the study tool in their local language (Hindi). After obtaining informed written consent, responses were collected and back-translated according to the WHO standards for translation. The WHOQOL-BREF questionnaire is available in Hindi and was used to collect information from the patients.
The data were collected by junior residents of the Department of Community and Family Medicine after training by the principal investigator regarding the administration of the study tool. Quality assurance of data was maintained with regular data entry checks and interim analysis by the principal investigator and co-investigators.
Biostatistical Analysis
The information collected was downloaded from Epicollect5 in Google Sheets and analyzed using jamovi (The jamovi project) [33]. Descriptive analyses were performed regarding the demographic and clinical profiles of CAM patients. Categorical variables such as sex, residence, education, occupation, vaccination status, and ICU requirement were expressed as proportions and percentages. Continuous variables such as age, QOL score, and improvement score were expressed as mean±standard deviation or median (interquartile range [IQR]) depending on the normality of the data. Continuous variables such as the SRI score were divided into 2 categories (good and poor) based on median score. A score of ≤7 was considered to indicate poor improvement. QOL was also categorized as good or poor based on the median score. A score of ≤52 was considered to indicate poor QOL. The median differences in the QOL score and SRI score across sociodemographic variables were assessed using the Mann-Whitney U-test. The associations between CAM severity, QOL category, and the clinical profile of CAM patients were assessed with the chi-square test. The mean differences between various domains of QOL and the clinical profile of CAM patients were assessed with the Student t-test. Multiple linear regression analysis was used to identify significant predictors of the QOL score of the patients with CAM, and an adjusted beta coefficient with a 95% confidence interval (CI) was determined. The QOL score, individual domains, and SRI score were correlated using Spearman correlation, and the correlation coefficient rho was calculated. Statistical significance was considered to be indicated by a p-value <0.05.
Ethics Statement
This study was approved by the institutional ethical committee of AIIMS Patna (No: AIIMS/Pat/IRC/2021/805). Ethical principles were adhered to throughout the study. Informed written consent was obtained for participation in the study.
Results
Of 120 patients who were admitted for treatment for CAM during the study period, 57 patients (47.5%) underwent follow-up, 20 (16.7%) did not consent to participate, 25 (20.8%) did not return for follow-up, and 18 (15.0%) died.
Sociodemographic Characteristics of CAM Patients
The mean±standard deviation age of the participants was 49.1±12.4 years. Approximately half (32, 52.5%), of the patients were 45 to 65 years old. Of the 61 total patients, the majority (41, 67.2%) were male, while only 10 of the patients (16.4%) were illiterate. Almost 70% of the patients (n=39) were employed, and 38 (63.3%) resided in rural areas (Table 1).
Clinical Details of CAM Patients
Among the 57 patients, 26 (45.6%) had at least one comorbidity. More than half of the total patients (36, 59.0%), had received both doses of the COVID-19 vaccine, while 13 (21.3%) had received no dose of any available COVID-19 vaccine.
Approximately two-thirds of the patients, 38 (66.7%), were in moderate to advanced stages of the disease process. Around 89.5% patients (n=51) received both medical and surgical treatment, and 28 patients (54.9%) who received both modes of treatment had good QOL, compared to 4 patients (66.7%) who received only medical treatment.
The median (IQR) duration of hospital stay for the CAM patients was approximately 25 days (3.0–35.0). Approximately 4 in 10 patients (23, 41.8%) were admitted to the ICU (Table 2) [34,35].
Association of Mucormycosis Stage with Sociodemographic Variables
Approximately 75% of the patients (21 patients) between 45 and 65 years of age had moderate to advanced disease. Around 57.9% of the men (22 patients) and 84.2% of the women (16 patients) had moderate to advanced CAM, and this sex difference in CAM stage was statistically significant (p=0.047). Additionally, 17 patients (54.8%) with good SRI and 21 patients (80.8%) with poor SRI had moderate to advanced disease. The difference in CAM stage according to improvement was also statistically significant (p=0.039) (Table 3).
SRI Scores of CAM Patients
The SRI scores of the patients are provided in Tables 1 and 3. Of a maximum total score of 9, the median (IQR) SRI score was 7 (6.0–8.0). The male patients had a median (IQR) score of 7 (6.0–8.0). Patients who had received intermediate-level education had a median score of 9 (6.5–9.0), while those with professional-level education had a median score of 4 (2.5–5.5). Statistically significant differences in scores were present between patients residing in urban and rural areas (7 [7–9] vs.(7 [7.0–9.0] vs. 6 [6.0–7.0], respectively; p=0.039) (Table 1). Approximately 54.4% of the patients (n=31) had good SRI. A statistically significant difference (p=0.039) was noted in the improvement score between the 2 stages of the disease, as almost three-fourths of the patients (14, 73.7%) with relatively early-stage disease showed good improvement, while more than half of the patients (21, 55.3%) with moderate to very advanced disease showed poor improvement (Table 3).
QOL of the Patients with CAM
The median (IQR) overall QOL score, of a total maximum score of 100, was 52 (41–63). The median (IQR) score was 50 (43–61) for the physical domain, 55 (35–65) for the psychological domain, 58 (42–67) for the social domain, and 50 (34–66) for the environmental domain (Figure 1).

Box-and-whisker plot showing median (interquartile range) quality of life (QOL) domain scores among patients with COVID-19–associated mucormycosis.
A statistically significant difference in patient QOL score was noted by occupation (p<0.001) and place of residence (p=0.015) (Table 1). Overall, 27 (47.4%; 95% CI, 34.9%–60.1%) of the 57 patients reported poor QOL. The difference in QOL categories across vaccination status, stages, modes of treatment, and ICU requirement are presented in Table 2. A statistically significant difference in QOL was found based on the presence or absence of any comorbidity (p<0.001). Despite having comorbidities, 20 affected patients (76.9%) had good QOL, while only 10 (32.3%; approximately one-third) patients with no comorbidity exhibited good improvement. Almost 56.3% of patients who did not require ICU admission and 43.5% of patients who required ICU admission had good QOL, but this difference was not significant (Table 2). Approximately half of patients (20, 50.0%) with poor SRI had relatively poor QOL. This difference was not significant (Table 2).
Clinical Profile of CAM Patients across QOL Domains and SRI Score
The domain-wise scores for QOL across various variables are given in Table 4.
Physical Domain
Relatively high physical domain scores were observed among CAM patients who had received the vaccine, who had comorbidities, who did not require ICU admission, who received surgical treatment, and who were in the early stage of the disease, but these differences lacked statistical significance (Table 4).
Psychological Domain
CAM patients with comorbidities had better psychological domain scores than those without, and this difference was statistically significant (p=0.023). Also, the psychological domain score was better among patients who had received COVID-19 vaccination, patients who did not require ICU admission, and patients with early-stage disease, although these differences were not statistically significant (Table 4).
Social Domain
CAM patients with comorbidities and those who had not received COVID-19 vaccination had relatively high social domain scores, and this difference was statistically significant (p<0.001). Social domain scores were also relatively high in patients who did not require ICU admission and patients in the early stage of the disease process, but these differences were not statistically significant (Table 4).
Environmental Domain
CAM patients with comorbidities exhibited better environmental domain scores than those without, and this difference was statistically significant (p<0.001). Environmental domain scores were relatively high in patients who had received the COVID-19 vaccination, who did not require ICU admission, and who were in the early stage of the disease process, but these differences were not statistically significant (Table 4).
Improvement Score
Relatively high SRI scores were observed among the patients with CAM who had received the vaccine and those who did not require ICU admission, and this difference was statistically significant (p=0.041 and p=0.016, respectively). SRI scores were also relatively high in patients with comorbidities and patients in the early stage of the disease, but these differences were not statistically significant (Table 4).
Associations of the QOL Score with Symptomology
Bivariate linear regression showed that facial pain (B, −8.19; 95% CI, −15.9 to −0.4), localized facial puffiness (B, 13.3; 95% CI, 2.74 to 23.9), and eye redness (B, −10.5; 95% CI, −17.8 to −3.5) were significantly associated with the QOL score (Table 5).
On multiple linear regression adjustment, regarding other symptoms, we found that headache (adjusted B, −12.3; 95% CI, −19.1 to −5.4), localized facial puffiness (adjusted B, −16.4; 95% CI, −26.6 to −6.3), facial discoloration (adjusted B, −23.4; 95% CI, −37.4 to −9.4), loosening of teeth (adjusted B, −18.7; 95% CI, −31.5 to −5.9), and facial palsy (adjusted B, −38.5; 95% CI, −65.8 to −11.2) were independently associated with the QOL score in patients with CAM (Table 5).
Correlations between the SRI Score and QOL Score Domains
We observed significant positive correlations between the physical (r=0.262, p=0.04) and psychological domains (r=0.447, p<0.001) of QOL and the SRI score. Regarding other domains of QOL, the social domain was negatively correlated and the environmental domain was positively correlated with the improvement score, but both correlations were statistically insignificant.
Discussion
With the onset of the COVID-19 pandemic, clinicians have seen an alarming increase in the number of CAM cases, reaching a level many times greater than the pre-COVID reported incidence of the disease [1]. The dual burden has impacted the health-related QOL. Although the prevalence of fungal coinfections in patients with COVID‐19 has been reported in many studies [24,34,36,37], our knowledge of the impact of mucormycosis on the QOL of these patients is very limited.
In this cross-sectional study, we surveyed 57 patients with CAM who underwent operation and engaged in follow-up during the study period. Approximately 15% of patients died before the 6-month follow-up. Another study in India reported around 50% 90-day mortality [38].
In this study, overall, nearly half of the patients (48%) with CAM had poor QOL. Some studies have shown that the impact of COVID-19 is itself a reason for poor QOL [39–41]. A separate study of patients with head and neck cancer who underwent treatment similar to CAM treatment also indicated poor QOL at the beginning of the therapy that persisted until the end of therapy, revealing the course of such debilitating diseases [42]. A study from Brazil showed poor QOL in patients with facial deformities relative to others [28].
In a study conducted by Pisulkar et al. [43], the mean global QOL after maxillectomy and rehabilitation was reported to be 54±22.9. In the present study, the median QOL was approximately 52, with a minimum of 41 and a maximum of 63.
An editorial by Ghosh et al. [36] comparing CAM presentation between India and other countries found that rhino-orbito-cerebral CAM presentation is most common in India, while pulmonary and disseminated CAM predominate in other countries. In the present study, we found that headache, loosening of teeth, localized facial puffiness, facial discoloration, and facial palsy independently predicted the QOL score of patients with CAM. Nonspecific clinical signs and symptoms, such as unilateral headaches and facial pain, fever, numbness, and nasal discharge, characterize the early phases of rhino-orbito-cerebral CAM [44]. In advanced cases, facial paralysis occurs [45]. Tooth loosening in the upper jaw and toothache are prompt signs for the early diagnosis of rhino-orbito-cerebral CAM [34].
The present study showed that 54.9% of patients with CAM who received both medical and surgical interventions had good QOL at follow-up. Combined medical and surgical treatment yields favorable outcomes [38]. A comparative study of India and other countries showed a similar result [46]. A study from Brazil, however, concluded no significant change in domain scores across treatment procedures [47].
In the present study, almost half of the CAM patients had at least one comorbidity; among them, nearly one-fourth had poor QOL. A study from Ranchi (Jharkhand, India) also showed poor QOL among those with comorbidities [48].
A study from North India [49] showed that morbidity and mortality rates were high among patients with post-tubercular mucormycosis, supporting our finding that the physical and environmental domains of QOL are affected more than other domains. These findings also align with a Turkish study on the QOL of patients with facial prostheses, a condition that is similarly debilitating to post-surgical mucormycosis. This explains the debilitating sequelae of mucormycosis that mainly affect these domains.
In our study, those who had been vaccinated for COVID-19 had better QOL than those who had not, indicating the effectiveness of the vaccine in decreasing COVID-19 severity [50–52] and the subsequent risk of CAM. Early recognition is the key to optimal treatment, improved outcomes, and improved QOL in patients with CAM [53].
Conclusion and Recommendations
Approximately 1 in 2 patients with CAM had poor QOL and poor SRI scores. The physical and environmental domains of QOL were most strongly affected.
Patients with comorbidities had relatively poor psychological, social, and environmental QOL domain scores, while patients who were not admitted to the ICU had relatively high improvement scores. Headache, localized puffiness of the face, loosening of the teeth, facial discoloration, and facial palsy were significantly associated with QOL score. We also observed weak positive correlations of the physical and psychological domains of QOL with the SRI score.
The highly invasive pathogenesis of mucormycosis often requires extensive surgical resection. Since the pandemic is not over and mucormycosis is not a preventable disease, it is imperative to rehabilitate these patients and reverse these effects. Both occupational and vocational rehabilitation (tertiary prevention) must be provided to ensure that patients who survive mucormycosis infection can continue to be functioning members of society.
Strengths and Limitations
Cases of mucormycosis, especially COVID-19–associated mucormycosis, are very rare, and the presence of a CAM epidemic during another pandemic (of COVID-19) is highly unusual. This study is one of the few studies in India and elsewhere to examine the QOL in mucormycosis-affected patients. The use of the validated WHO-BREF scale for assessing QOL is another strength of the study. We assessed the contribution of each symptom to overall QOL in patients with CAM.
This study is not without limitations. First, a mixed-methods approach would have provided better insight into QOL but was not feasible, as many patients were in a debilitating condition. Second, the sample size used was relatively small, but since this is a rare condition and the study was performed under epidemic conditions, we consider the sample size to be reasonable in context. Third, we could not capture the baseline QOL and thus could not compare it with the follow-up value.
HIGHLIGHTS
• Mucormycosis and COVID-19–associated mucormycosis (CAM) are relatively rare worldwide. The COVID-19 pandemic has led to high morbidity and mortality due to the nature of the disease and has compromised the quality of life (QOL) of patients who have recovered from CAM.
• This study documents the QOL of patients with CAM at a 6-month follow-up after interventions.
• The use of the validated abbreviated World Health Organization Quality of Life questionnaire (WHOQOL BREF) is an important highlight of this study.
• Approximately 1 in 2 patients with CAM showed poor QOL. The importance of rehabilitation for such recovered patients is highlighted in this study.
Notes
Ethics Approval
This study was approved by the Institute Ethics Committee of AIIMS, Patna (No: AIIMS/Pat/IEC/2021/805). We adhered to the principles of ethics throughout the study and thereafter and performed the study in accordance with the principles of the Declaration of Helsinki. Only those who provided written informed consent were included.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding
None.
Availability of Data
The datasets are not publicly available, but they are available from the corresponding author upon reasonable request.
Authors’ Contributions
Conceptualization: PK, SA, KB; Data curation: RRUR, NR, DA; Data analysis: RRUR, NR, DA; Methodology: PK, SA, RRUR; Supervision: PK, SA, KB; Writing-original draft: RRUR, NR; Writing-review & editing: all authors. All authors read and approved the final manuscript.
Acknowledgements
We would like to acknowledge Dr. Akanksha Yadav, junior resident, and intern doctors from the Department of Community and Family Medicine at AIIMS Patna for their contribution to the study.