Articles
- Page Path
- HOME > Osong Public Health Res Perspect > Volume 6(6); 2015 > Article
-
Original Article
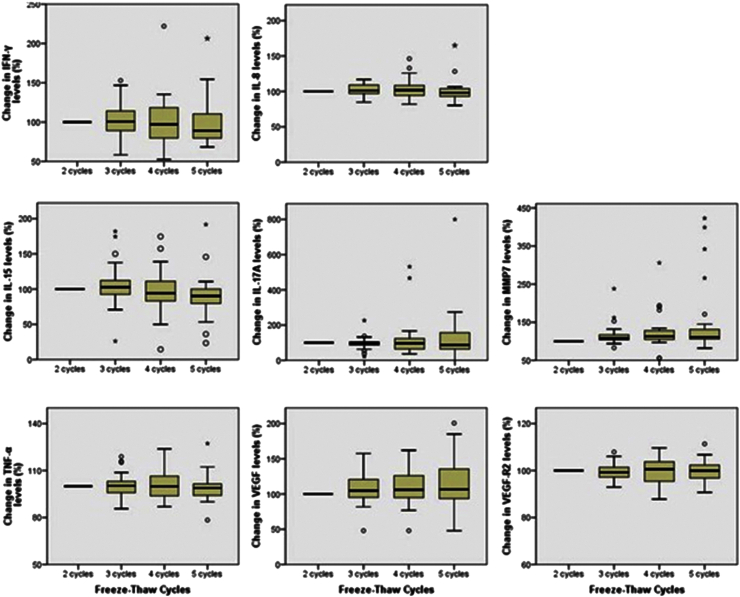
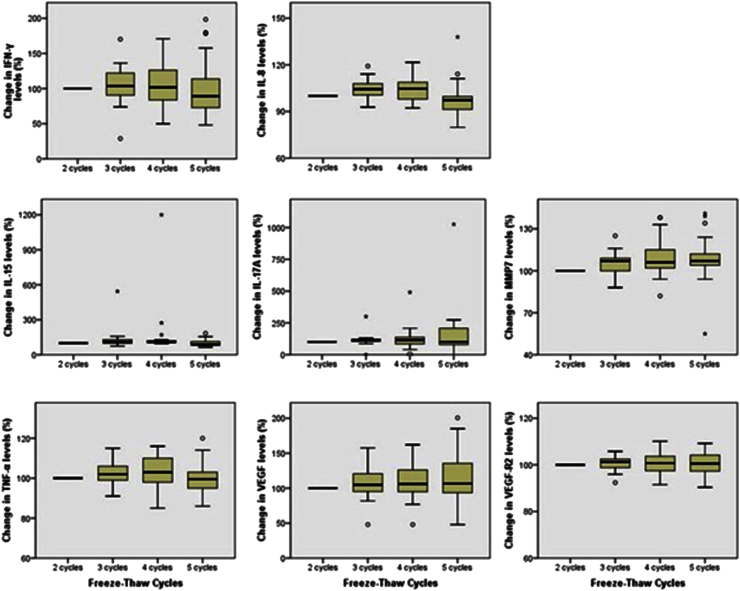
Effect of Repeated Freezing and Thawing on Biomarker Stability in Plasma and Serum Samples - Jae-Eun Leea, Shine Young Kimb, So-Youn Shina
-
Osong Public Health and Research Perspectives 2015;6(6):357-362.
DOI: https://doi.org/10.1016/j.phrp.2015.11.005
Published online: November 26, 2015
aNational Biobank of Korea, Center for Genome Sciences, Korea National Institute of Health, Korea Centers for Disease Control and Prevention, Chungcheongbuk-do, Korea
bDepartment of Laboratory Medicine, Pusan National University School of Medicine, Busan, Korea
- ∗Corresponding author. leukoso@hanmail.net
• Received: September 24, 2015 • Accepted: November 7, 2015
Copyright © 2015 Korea Centers for Disease Control and Prevention. Published by Elsevier Korea LLC. All rights reserved.
This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
Figure & Data
References
Citations
Citations to this article as recorded by 

- Reliability, stability during long-term storage, and intra-individual variation of circulating levels of osteopontin, osteoprotegerin, vascular endothelial growth factor-A, and interleukin-17A
Tomoki Nakamizo, John Cologne, Takeshi Kishi, Tetsuya Takahashi, Mayumi Inoue, Hiroyuki Ryukaku, Tomonori Hayashi, Yoichiro Kusunoki, Saeko Fujiwara, Waka Ohishi
European Journal of Medical Research.2024;[Epub] CrossRef - Biobanking for Neurodegenerative Diseases: Challenge for Translational Research and Data Privacy
Emilia Giannella, Valentino Notarangelo, Caterina Motta, Giulia Sancesario
The Neuroscientist.2023; 29(2): 190. CrossRef - Keep cool! Observed temperature variations at different process stages of the biobanking workflow – examples from the Leipzig medical biobank
Juliane Weikert, Angelina Mehrländer, Ronny Baber
Journal of Laboratory Medicine.2023; 47(2): 69. CrossRef - The Stability of the Anti-Müllerian Hormone in Serum and Plasma Samples under Various Preanalytical Conditions
Radana Vrzáková, Václav Šimánek, Ondřej Topolčan, Vladimír Vurm, David Slouka, Radek Kučera
Diagnostics.2023; 13(8): 1501. CrossRef - Differences in the inflammatory proteome of East African and Western European adults and associations with environmental and dietary factors
Godfrey S Temba, Nadira Vadaq, Vesla Kullaya, Tal Pecht, Paolo Lionetti, Duccio Cavalieri, Joachim L Schultze, Reginald Kavishe, Leo AB Joosten, Andre J van der Ven, Blandina T Mmbaga, Mihai G Netea, Quirijn de Mast
eLife.2023;[Epub] CrossRef - Influence of Freezing Temperature, Freezing Duration, and Repeated Freeze/Thaw Cycles on Electrophoretic Profiles in the White Stork (Ciconia ciconia)
Milan Thorel, Yannick Roman, Antoine Leclerc
Journal of Avian Medicine and Surgery.2023;[Epub] CrossRef - Clinical Spectroscopy: Lost in Translation?
James M. Cameron, Christopher Rinaldi, Samantha H. Rutherford, Alexandra Sala, Ashton G. Theakstone, Matthew J. Baker
Applied Spectroscopy.2022; 76(4): 393. CrossRef - Correlation of Repeat Measurements of 27 Candidate Protein Markers for Colorectal Cancer Screening Taken Three Years and Multiple Freeze–Thaw Cycles Apart
Megha Bhardwaj, Petra Schrotz-King, Hermann Brenner
Life.2022; 12(3): 359. CrossRef - Development of quinone linked immunosorbent assay (QuLISA) based on using Folin’s reagent as a non-enzymatic tag: Application to analysis of food allergens
Mahmoud El-Maghrabey, Yudai Sato, Fatema Kaladari, Naoya Kishikawa, Naotaka Kuroda
Sensors and Actuators B: Chemical.2022; 368: 132167. CrossRef - A systematic review and meta-analysis of preanalytical factors and methodological differences influencing the measurement of circulating vascular endothelial growth factor
Ulrika Sjöbom, Anders K. Nilsson, Hanna Gyllensten, Ann Hellström, Chatarina Löfqvist, Angela J. Glading
PLOS ONE.2022; 17(7): e0270232. CrossRef - Cytokines: From Clinical Significance to Quantification
Chao Liu, Dewei Chu, Kourosh Kalantar‐Zadeh, Jacob George, Howard A. Young, Guozhen Liu
Advanced Science.2021;[Epub] CrossRef - Microheterogeneity and preanalytical stability of protein biomarkers of inflammation and renal function
Jie Gao, Arve Ulvik, Adrian McCann, Per Magne Ueland, Klaus Meyer
Talanta.2021; 223: 121774. CrossRef - The effect of pre-analytical handling on the stability of fractalkine, monocyte chemoattractant protein 1 (MCP1), interleukin 6 and interleukin 8 in samples of human cerebrospinal fluid
Winnie Charlotte Pedersen Mortensen, Laila Bendix, Hanne Irene Jensen, Claus Varnum, Lasse Enkebølle Rasmussen, Jørgen T. Lauridsen, Nis Borbye-Lorenzen, Kristin Skogstrand, Palle Toft, Henrik Bjarke Vaegter, Morten Rune Blichfeldt-Eckhardt
Journal of Immunological Methods.2021; 494: 113057. CrossRef - Standardizing salivary lactoferrin measurements to obtain a robust diagnostic biomarker for Alzheimer's disease
Fernando Bartolome, Gorka Orive, Eva Carro
Alzheimer's & Dementia: Diagnosis, Assessment & Di.2021;[Epub] CrossRef - Pre-analytical considerations in biomarker research: focus on cardiovascular disease
Elena Revuelta-López, Jaume Barallat, Adriana Cserkóová, Carolina Gálvez-Montón, Allan S. Jaffe, James L. Januzzi, Antoni Bayes-Genis
Clinical Chemistry and Laboratory Medicine (CCLM).2021; 59(11): 1747. CrossRef - Vaginal cytokine profile and microbiota before and after lubricant use compared with condomless vaginal sex: a preliminary observational study
Susan Tuddenham, Christina A. Stennett, Richard A. Cone, Jacques Ravel, Andrew N. Macintyre, Khalil G. Ghanem, Xin He, Rebecca M. Brotman
BMC Infectious Diseases.2021;[Epub] CrossRef - Proximity Extension Assay in Combination with Next-Generation Sequencing for High-throughput Proteome-wide Analysis
Lotta Wik, Niklas Nordberg, John Broberg, Johan Björkesten, Erika Assarsson, Sara Henriksson, Ida Grundberg, Erik Pettersson, Christina Westerberg, Elin Liljeroth, Adam Falck, Martin Lundberg
Molecular & Cellular Proteomics.2021; 20: 100168. CrossRef - Serum or plasma? An old question looking for new answers
Mario Plebani, Giuseppe Banfi, Sergio Bernardini, Francesco Bondanini, Laura Conti, Romolo Dorizzi, Fulvio Enrico Ferrara, Rita Mancini, Tommaso Trenti
Clinical Chemistry and Laboratory Medicine (CCLM).2020; 58(2): 178. CrossRef - Association Between Intravenous Fluid Bolus and Biomarker Trajectory During Prehospital Care
Emily B. Brant, Jason Kennedy, Christian Martin-Gill, Vanessa Jackson, Octavia M. Peck Palmer, Clifton W. Callaway, Jeremy M. Kahn, Donald M. Yealy, Derek C. Angus, Christopher W. Seymour
Prehospital Emergency Care.2020; 24(2): 196. CrossRef - Evaluating sample stability in the clinical laboratory with the help of linear and non-linear regression analysis
Joachim K.W. Pum
Clinical Chemistry and Laboratory Medicine (CCLM).2020; 58(2): 188. CrossRef - Optimization of Saliva Collection and Immunochromatographic Detection of Salivary Pepsin for Point-of-Care Testing of Laryngopharyngeal Reflux
Young Ju Lee, Jiyoon Kwon, Sanggyeong Shin, Young-Gyu Eun, Jae Ho Shin, Gi-Ja Lee
Sensors.2020; 20(1): 325. CrossRef - Stability of 12 T-helper cell-associated cytokines in human serum under different pre-analytical conditions
Ulrik Stervbo, Sharon Bajda, Patrizia Wehler, Benjamin J. Rohn, Melanie Streichhahn, Sehriban Temizsoy, Eva Kohut, Toralf Roch, Richard Viebahn, Timm H. Westhoff, Nina Babel
Cytokine.2020; 129: 155044. CrossRef - Quinone-based antibody labeling reagent for enzyme-free chemiluminescent immunoassays. Application to avidin and biotinylated anti-rabbit IgG labeling
Mahmoud El-Maghrabey, Naoya Kishikawa, Shiori Harada, Kaname Ohyama, Naotaka Kuroda
Biosensors and Bioelectronics.2020; 160: 112215. CrossRef - Standardized approaches for clinical sampling and endpoint ascertainment in tuberculous meningitis studies
Ursula K Rohlwink, Felicia C Chow, Sean Wasserman, Sofiati Dian, Rachel PJ Lai, Lidya Chaidir, Raph L Hamers, Robert J Wilkinson, David R Boulware, Fiona V Cresswell, Arjan van Laarhoven
Wellcome Open Research.2020; 4: 204. CrossRef - Exploring pre-analytical factors for the optimisation of serum diagnostics: Progressing the clinical utility of ATR-FTIR spectroscopy
James M. Cameron, Holly J. Butler, David J. Anderson, Loren Christie, Lily Confield, Katie E. Spalding, Duncan Finlayson, Stuart Murray, Zanib Panni, Christopher Rinaldi, Alexandra Sala, Ashton G. Theakstone, Matthew J. Baker
Vibrational Spectroscopy.2020; 109: 103092. CrossRef - QuantiFERON-TB Gold Plus with Chemiluminescence Immunoassay: Do We Need a Higher Cutoff?
Francesco Bisognin, Giulia Lombardi, Maria Carla Re, Paola Dal Monte, Geoffrey A. Land
Journal of Clinical Microbiology.2020;[Epub] CrossRef - Stability of serum biochemical markers during standard long-term storage and with a single thawing
V. A. Kozlova, V. A. Metelskaya, M. S. Pokrovskaya, I. A. Efimova, O. A. Litinskaya, V. A. Kutsenko, E. B. Yarovaya, S. A. Shalnova, O. M. Drapkina
Cardiovascular Therapy and Prevention.2020; 19(6): 2736. CrossRef - Development and application of a porcine specific ELISA for the quantification of soluble CD163
J. Alex Pasternak, Daniel J. MacPhee, John C.S. Harding
Veterinary Immunology and Immunopathology.2019; 210: 60. CrossRef - Dynamics of circulating TNF during adalimumab treatment using a drug-tolerant TNF assay
Lea C. Berkhout, Merel J. l’Ami, Jill Ruwaard, Margreet H. Hart, Pleuni Ooijevaar-de Heer, Karien Bloem, Michael T. Nurmohamed, Ronald F. van Vollenhoven, Maarten Boers, Daniel F. Alvarez, Catherine H. Smith, Gerrit J. Wolbink, Theo Rispens
Science Translational Medicine.2019;[Epub] CrossRef - Standardized approaches for clinical sampling and endpoint ascertainment in tuberculous meningitis studies
Ursula K Rohlwink, Felicia C Chow, Sean Wasserman, Sofiati Dian, Rachel PJ Lai, Lidya Chaidir, Raph L Hamers, Robert J Wilkinson, David R Boulware, Fiona V Cresswell, Arjan van Laarhoven
Wellcome Open Research.2019; 4: 204. CrossRef - The Effect of Anticoagulants, Temperature, and Time on the Human Plasma Metabolome and Lipidome from Healthy Donors as Determined by Liquid Chromatography-Mass Spectrometry
Manoj Khadka, Andrei Todor, Kristal M. Maner-Smith, Jennifer K. Colucci, ViLinh Tran, David A. Gaul, Evan J. Anderson, Muktha S. Natrajan, Nadine Rouphael, Mark J. Mulligan, Circe E. McDonald, Mehul Suthar, Shuzhao Li, Eric A. Ortlund
Biomolecules.2019; 9(5): 200. CrossRef - A caspase-6-cleaved fragment of Glial Fibrillary Acidic Protein as a potential serological biomarker of CNS injury after cardiac arrest
Ditte S. Jonesco, Christian Hassager, Martin Frydland, Jesper Kjærgaard, Morten Karsdal, Kim Henriksen, Stephen D. Ginsberg
PLOS ONE.2019; 14(11): e0224633. CrossRef - Angiogenic Factor and Cytokine Analysis among Patients Treated with Adjuvant VEGFR TKIs in Resected Renal Cell Carcinoma
Wenxin Xu, Maneka Puligandla, Judith Manola, Andrea J. Bullock, Daniel Tamasauskas, David F. McDermott, Michael B. Atkins, Naomi B. Haas, Keith Flaherty, Robert G. Uzzo, Janice P. Dutcher, Robert S. DiPaola, Rupal S. Bhatt
Clinical Cancer Research.2019; 25(20): 6098. CrossRef - Stability of important antibodies for kidney disease: pre-analytic methodological considerations
Qiuxia Han, Songyan Li, Bo Fu, Dongwei Liu, Maoqing Wu, Xiaoli Yang, Guangyan Cai, Zhangsuo Liu, Xiangmei Chen, Hanyu Zhu
PeerJ.2018; 6: e5178. CrossRef - The association of angiogenic factors and chronic kidney disease
Christopher E. Anderson, L. Lee Hamm, Gem Batuman, Damodar R. Kumbala, Chung-Shiuan Chen, Swapna G. Kallu, Ravi Siriki, Shilpa Gadde, Myra A. Kleinpeter, N. Kevin Krane, Eric E. Simon, Jiang He, Jing Chen
BMC Nephrology.2018;[Epub] CrossRef - Strong impact on plasma protein profiles by precentrifugation delay but not by repeated freeze-thaw cycles, as analyzed using multiplex proximity extension assays
Qiujin Shen, Johan Björkesten, Joakim Galli, Daniel Ekman, John Broberg, Niklas Nordberg, Annika Tillander, Masood Kamali-Moghaddam, Gunnel Tybring, Ulf Landegren
Clinical Chemistry and Laboratory Medicine (CCLM).2018; 56(4): 582. CrossRef - Plasma-Derived Inflammatory Proteins Predict Oral Squamous Cell Carcinoma
Kelly Yi Ping Liu, Xian Jun David Lu, Yuqi Sarah Zhu, Nhu Le, Hugh Kim, Catherine F. Poh
Frontiers in Oncology.2018;[Epub] CrossRef - Is vascular endothelial growth factor a useful biomarker in giant cell arteritis?
Nicola Goodfellow, Julien Morlet, Surjeet Singh, Afsie Sabokbar, Andrew Hutchings, Vanshika Sharma, Jana Vaskova, Shauna Masters, Allahdad Zarei, Raashid Luqmani
RMD Open.2017; 3(1): e000353. CrossRef - Indices of immune function used by ecologists are mostly unaffected by repeated freeze-thaw cycles and methodological deviations
Arne Hegemann, Sara Pardal, Kevin D. Matson
Frontiers in Zoology.2017;[Epub] CrossRef - The Enhanced Liver Fibrosis (ELF) Panel: Analyte Stability Under Common Sample Storage Conditions Used in Clinical Practice
Oliver J Kennedy, Julie Parkes, Sudeep Tanwar, Paul M Trembling, William M Rosenberg
The Journal of Applied Laboratory Medicine.2017; 1(6): 720. CrossRef - Instability of Plasma and Serum Progastrin-Releasing Peptide During Repeated Freezing and Thawing
Jae-Eun Lee, Jin-Hyun Lee, Maria Hong, Seul-Ki Park, Ji-In Yu, So-Youn Shin, Shine Young Kim
Osong Public Health and Research Perspectives.2016; 7(6): 351. CrossRef - Effects of Long-Term Storage Time and Original Sampling Month on Biobank Plasma Protein Concentrations
Stefan Enroth, Göran Hallmans, Kjell Grankvist, Ulf Gyllensten
EBioMedicine.2016; 12: 309. CrossRef


 PubReader
PubReader Cite
Cite
