Articles
- Page Path
- HOME > Osong Public Health Res Perspect > Volume 12(5); 2021 > Article
-
Review Article
The role of lipids in the pathophysiology of coronavirus infections -
Milad Zandi1,2
 , Parastoo Hosseini1,2
, Parastoo Hosseini1,2 , Saber Soltani1,2
, Saber Soltani1,2 , Azadeh Rasooli3
, Azadeh Rasooli3 , Mona Moghadami4
, Mona Moghadami4 , Sepideh Nasimzadeh5
, Sepideh Nasimzadeh5 , Farzane Behnezhad1
, Farzane Behnezhad1
-
Osong Public Health and Research Perspectives 2021;12(5):278-285.
DOI: https://doi.org/10.24171/j.phrp.2021.0153
Published online: October 15, 2021
1Department of Virology, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran
2Research Center for Clinical Virology, Tehran University of Medical Sciences, Tehran, Iran
3Department of Biochemistry, Faculty of Sciences, Payame Noor University, Tehran, Iran
4Department of Medical Biotechnology, School of Medicine, Babol University of Medical Sciences, Babol, Iran
5Department of Virology, School of Medicine, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
- Corresponding author: Milad Zandi Department of Virology, School of Public Health, Tehran University of Medical Sciences, Poursina Avenue, Qods Street, Enqelab Square, Tehran, Iran E-mail: Miladzandi416@gmail.com
- Milad Zandi and Parastoo Hosseini contributed equally as co-first authors.
© 2021 Korea Disease Control and Prevention Agency.
This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
- Abstract
- Graphical abstract
- Introduction
- Lipids and Viral Infections
- The Genome of Coronaviruses
- Structural Proteins of Coronaviruses
- Lipids and Coronavirus Entry
- Lipids and Proliferation of Coronaviruses
- Lipid Pathways as Potential Therapeutic Targets in HCoV Infections
- Conclusion
- Article information
- References
Abstract
- Coronaviruses, which have been known to cause diseases in animals since the 1930s, utilize cellular components during their replication cycle. Lipids play important roles in viral infection, as coronaviruses target cellular lipids and lipid metabolism to modify their host cells to become an optimal environment for viral replication. Therefore, lipids can be considered as potential targets for the development of antiviral agents. This review provides an overview of the roles of cellular lipids in different stages of the life cycle of coronaviruses.
- Coronaviruses are a group of viruses that belong to the Coronaviridae family. This viral family is subdivided into 4 genera, including alpha-coronaviruses, beta-coronaviruses, gamma-coronaviruses, and delta-coronaviruses [1]. Human coronaviruses (HCoVs) belong to the alpha-coronavirus or beta-coronavirus genera. Although HCoVs generally cause mild to severe respiratory diseases [2], some coronaviruses have evolved to cross the species barrier [3], giving rise to diseases such as severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome coronavirus (MERS-CoV), which caused viral outbreaks in 2003 and 2012, respectively [4,5].
- In late December 2019, a new member of Coronaviridae family, named SARS-CoV-2, was discovered in China [6]. SARS-CoV-2, like SARS-CoV and MERS-CoV, is a zoonotic virus that has crossed the species barrier. Thus, SARS-CoV-2 is an emerging virus, and it causes coronavirus disease 2019 (COVID-19) [7,8]. As of September 14, 2021, the World Health Organization has reported more than 200 million confirmed cases of COVID-19, including more than 4 million deaths [9].
- Coronaviruses have a positive-strand RNA enclosed in a protein shell that is surrounded by a host cell-derived membrane [10]. Coronaviruses cross the host cell membrane at least twice in the cell entry and exit stages of viral particles during the replication cycle. Replication of coronaviruses depends on their interactions with cellular lipids, which are essential for the successful life cycle of coronaviruses [7,11].
- Cellular lipids play key roles in viral infections, particularly during the entry process and virion maturation. Lipid signaling and its synthesis machinery in host cells can be remodeled via proteins encoded by viruses [12]. Moreover, viruses alter cellular metabolism to promote proficient viral replication [13]. In this paper, we review the interactions of coronaviruses with the cellular lipids required for an effective viral life cycle.
Introduction
- Viruses have complex interactions with cells. According to recent studies, cellular lipids play important roles in the viral life cycle, such as viral-to-host cell membrane fusion, viral replication, and endocytosis and exocytosis [12]. Viral entry involves specific lipids, which vary among viruses [14]. The combination of lipids and proteins in the host cell membrane and the viral envelope plays a key role in viral infections [10]. In fact, viruses can alter the metabolism and signaling of lipids in host cells in order to facilitate their replication, and such viral interactions with cellular lipids have shown to be different among viruses. [15]. The pathways of cellular lipid biogenesis are among the most important cellular pathways hijacked by viruses. Lipids play a crucial role in the formation of viral replication organelles, as well as energy production for viral replication. Lipids are also important for regulating the proper cellular arrangement of viral proteins and the assembly, trafficking, and release of viral particles [16–18].
- Lipid rafts are specialized microdomains (10 to 200 nm) of the cell membrane that are found on the membranes of endosomes and exosomes in the endoplasmic reticulum (ER) and the Golgi complex [19]. These microdomains contain sphingolipids, cholesterol, various receptors, and other proteins [20]. Lipid rafts play important roles in viral infection, for instance in endocytosis or during different stages of pathogenesis [21]. A vital component of lipid rafts is cholesterol, which plays a major role in viral entry and release for enveloped viruses such as coronaviruses and influenza virus [22]. The major surface glycoproteins of influenza virus, hemagglutinin (HA) and neuraminidase, are associated with lipid rafts, and depletion of cholesterol by methyl-β-cyclodextrin (MβCD) can reduce the transport of HA from the trans-Golgi network to the cell membrane [23]. The requirement of cholesterol for viral pathogenesis may differ in respiratory RNA viruses, and the depletion of cholesterol by MβCD is involved in the increased budding of influenza A virus (IAV) from the host cells during pathogenesis [24]. In fact, cholesterol is essential as a vital component for sustaining IAV and respiratory syncytial virus (RSV) infectivity [25].
- In a previous study, pretreatment of influenza virions with MβCD efficiently depleted cholesterol in the envelope and considerably reduced the infectivity of the virus in a dose-dependent manner [15]. In addition, the depletion of cholesterol by MβCD decreased RSV infection and interrupted lipid raft microdomains, implying that cholesterol in lipid rafts is essential for the interactions of viral proteins during infection [25]. According to prior research, human rhinovirus serotype 2 can enter into the cell through clathrin-mediated endocytosis, and the depletion of cholesterol by MβCD can prevent clathrin-mediated endocytosis and decrease viral entry [26]. Some studies showed that ceramide-rich platforms play key roles in rhinovirus infections. This suggests a novel target to treat rhinovirus infections [27]. For hepatitis C virus (HCV), it is clear that the utilization of very low-density lipoprotein secretion machinery during HCV infection supports the exit of the virus from its cellular host [28].
Lipids and Viral Infections
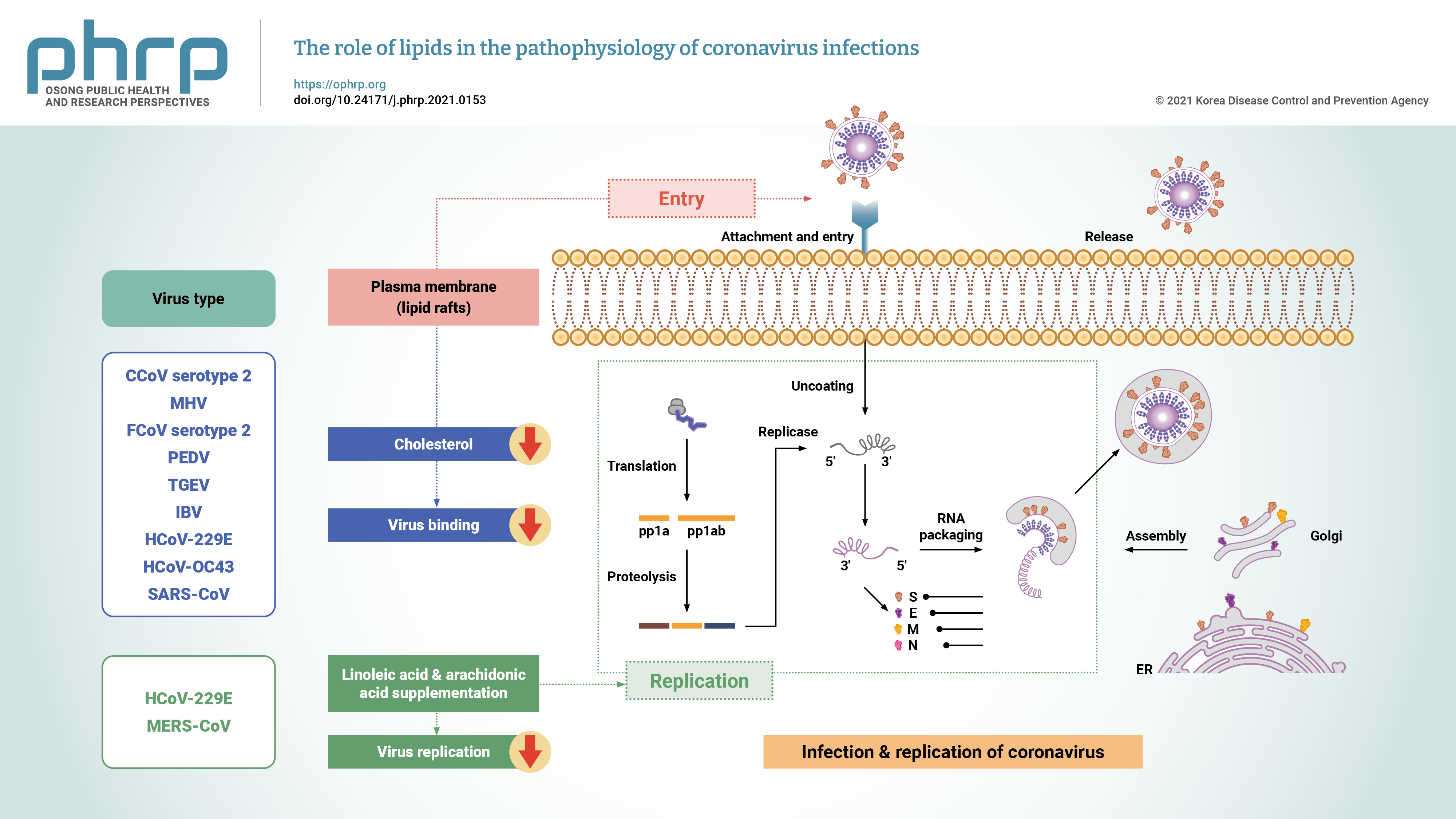
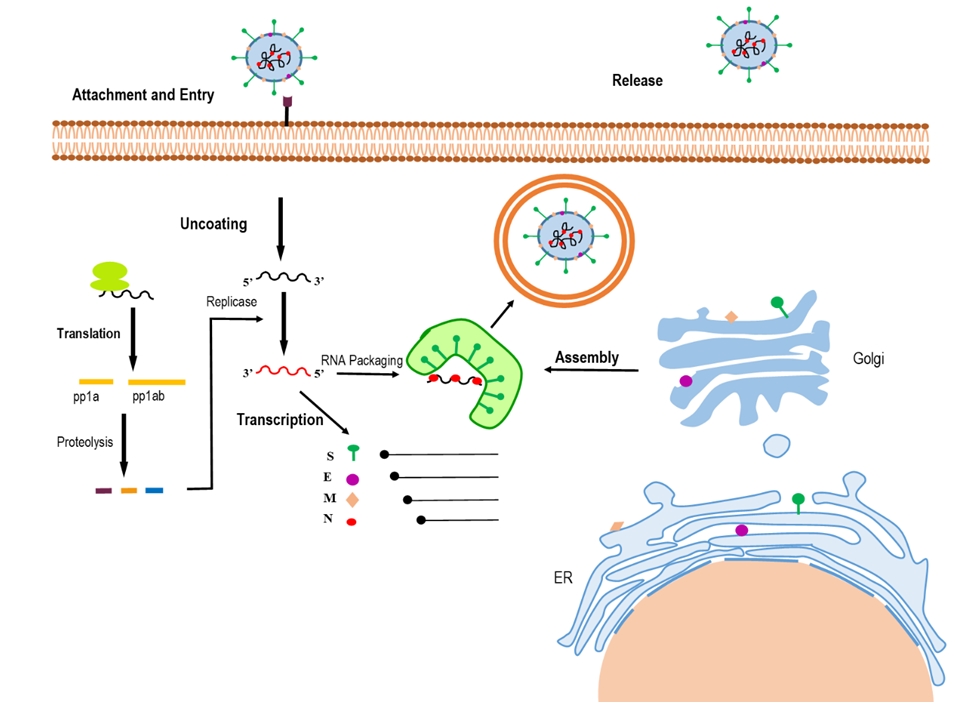
- Coronaviruses are enveloped positive-stranded RNA viruses with a genome size of about 27 to 30 kb, 5′‐cap structure, and 3′‐poly-A tail [29]. The genome of coronaviruses contains several segments—including untranslated regions (UTRs), the spike (S) protein, the envelope (E) protein, the membrane (M) protein, and the nucleocapsid (N) protein—organized as follows: 5′-leader-UTR-replicase-S-E-M-N-3′UTR-poly(A) tail [30,31]. There are 2 overlapping open reading frames (ORFs), referred to as ORF1a and ORF1b, in the replicase gene. These ORFs encode 2 polypeptides (pp1a and pp1ab), which are processed into 16 nonstructural proteins by viral-encoded enzymes including 3-chymotrypsin‐like protease or main protease and 1 or 2 papain‐like proteases (Figure 1) [31,32].
The Genome of Coronaviruses
- The surface spike proteins of coronaviruses are formed by trimers of S molecules. The S protein is a class I viral fusion protein that plays a major role in viral binding to cellular receptors in order to enter the host cell [33]. The S protein undergoes modification by N-linked glycosylation in the ER [34]. The S glycoprotein contains 2 subunits (S1 and S2). The S1 subunit is variable, whereas the S2 subunit is conserved among diverse isolates of a single coronavirus. The S1 subunit is responsible for the binding of the virus to the cell receptor, whereas S2 mediates the fusion of the viral envelope and cellular membranes [35]. The M protein, which is considered to be the most abundant structural protein in coronaviruses, is N-glycosylated [36], while some beta-coronaviruses are modified by O-linked glycosylation [37]. The glycosylation of the M protein influences the interferon-inducing ability of some coronaviruses and also shapes the virion envelope [36]. The E protein is the smallest protein and is found in virions in limited amounts. Furthermore, the E protein is essential for viral infectivity and also plays a key role in virion assembly [38]. Some studies have shown that the E protein plays a role in viral pathogenesis [39]. The N protein, which is the only protein located in the ribonucleoprotein core, is made up of 3 domains, including the N-terminal domain and C-terminal domain, which are separated by an intrinsically disordered central region. The N protein is a phosphoprotein that binds to the RNA genome and is involved in the formation of the nucleocapsid [40]. In addition, HA esterase as a structural protein is encoded by beta-coronaviruses of lineage A, such as OC43-CoV. During viral infection, viruses can alter host cell metabolism and reprogram it to use cellular factors [13].
Structural Proteins of Coronaviruses
- To initiate infection, coronaviruses need to enter into the cell through interactions between the viral spike proteins and the cellular receptors located on the surface of the host cell [33]. The cellular plasma membrane contains subdomains of lipid rafts composed of cholesterol and glycosphingolipids [41]. SARS-CoV-2 and SARS-CoV use the angiotensin-converting enzyme-2 (ACE2) receptor for cellular entry [42]. Interactions between the ACE2 receptor and the spike protein are facilitated by cholesterol-rich microdomains. Studies have shown that the depletion of cholesterol by MβCD in cells expressing ACE2 leads to a decrease in binding viral S glycoproteins in 50% of SARS-CoV infections. Therefore, MβCD affects cholesterol levels and ACE2 receptor expression. The depletion of cholesterol by MβCD also prevents the attachment of coronaviruses to the cell membrane [43]. Human coronavirus 229E (HCoV-229E) binds to the cellular receptor (aminopeptidase N or cluster of differentiation 13 [CD13]) for cellular infection, CD13 localized in lipid rafts, so the depletion of cholesterol by MβCD decreases the likelihood of HCoV-229E infection and prevents viral entry into host cells [44]. According to in vitro experiments, cholesterol supplementation enhances the propensity of the virus to fuse with the cell membrane. Clathrins, caveolins, and dynamin in lipid rafts play an important role in viral entry [43]. Some coronaviruses utilize lipid rafts for the cellular entry process. It has been reported that lipid rafts are required in the attachment process during infectious bronchitis virus infection [45], and another study also showed that lipid rafts are required for the entry of SARS-CoV into Vero E6 cells [46].
- Cholesterol in the plasma membrane of target cells is also important for SARS-CoV infection [47]. It has been reported that drugs causing cholesterol depletion can inhibit the entry of murine hepatitis virus and HCoV-229E into host cells [48]. Since coronaviruses are enveloped particles, fusion with the host cell membrane is necessary before the internalization of viral particles into cells [49], SARS-CoV uses various endocytic routes including clathrin-mediated dependent, lipid raft-dependent, and clathrin- and caveolae-independent endocytosis [50]. However, HCoVs such as human coronavirus OC43 (HCoV-OC43) use caveolae-dependent endocytosis as the entry pathway [51] and feline infectious peritonitis virus uses clathrin-independent and caveolin-independent endocytosis to enter the host cell (Table 1) [33,44–46,48–60]. Therefore, it is necessary to investigate the entry pathways of coronaviruses and the mechanisms of those pathways in order to design selective inhibitors for the entry stage of coronaviruses.
Lipids and Coronavirus Entry
- The replication of most positive-strand RNA viruses occurs in the cytoplasm of the host cell. These viruses induce the formation of subcellular membranes known as virus factories or viroplasm, where they can efficiently replicate, recruit host components, and escape from the defense mechanisms of host cells [61,62].
- Depending on the viral family and genus, these remodeled intracellular membranes may originate from various organelles, including the ER, late endosomes/lysosomes, or the mitochondrial outer membrane. Positive-sense RNA viruses induce the formation of 2 types of vesicles: (1) spherules, which are generated by viruses in the Togaviridae, Bromoviridae, and Nodaviridae families; and (2) double-membrane vesicles (DMVs), generated by the Coronaviridae, Arteriviridae, and Picornaviridae families [63,64].
- Although the role of DMVs has not been fully clarified, they may act as autophagosomes in autophagy. The virus uses DMVs to protect against host antiviral responses or in viral replication [65]. DMV formation requires both viral and host factors. In coronaviruses, the nonstructural proteins nsp3, nsp4, and nsp6, which contain predicted transmembrane domains, play an important role in DMV biogenesis [66].
- The replication process of coronaviruses that occurs in the cytoplasm of the infected host cells is similar to that of other positive-strand RNA viruses and is associated with intracellular lipid membranes derived from various organelles. Moreover, coronaviruses can also utilize cellular lipids for their replication, and thus the replication of viruses induces cellular membrane remodeling [67]. HCoV-229E infection can rearrange the cellular lipid profile, and lysophosphatidylcholine, lysophosphatidylethanolamine, and fatty acids are upregulated after infection with HCoV-229E. However, HCoV-229E replication is suppressed by exogenous supplementation of linoleic acid (LA) or arachidonic acid (AA) in infected cells. LA and AA have potent modulatory effects on MERS-CoV infection and replication of HCoV-229E [53].
- The nonstructural proteins nsp3, nsp4, and nsp6 facilitate the formation of replication/transcription complexes by inducing the formation of DMVs [68]. Some cellular enzymes such as cytosolic phospholipase A2α are involved in the formation of DMVs, which take part in the replication of coronaviruses [69]. As described above, the development of new inhibitors could play a strategic role in preventing virus transmission from infected individuals to the healthy population.
Lipids and Proliferation of Coronaviruses
- Since lipids play vital roles in the viral life cycle, using drugs that can target lipid metabolism may therefore interfere with infections of SARS-CoV-2 and other coronaviruses [70]. Phytosterols can affect viral infection by decreasing the levels of cholesterol in cell membrane [71]. In this regard, umifenovir and chloroquine are antiviral drugs that inhibit the process of endocytosis [72]. The mechanism of actions of these antiviral drugs suggests the significance of the viral membrane for developing potent drugs. Statins can reduce cholesterol levels and disrupt lipid rafts, thereby inhibiting coronavirus infection [73].
Lipid Pathways as Potential Therapeutic Targets in HCoV Infections
- Overall, the replication process of coronaviruses relies on cellular lipids, and these viruses alter the cellular lipid profile. Coronaviruses can stimulate membrane lipid remodeling in host cells and utilize cellular lipids to form viral particles or viral replication complexes, which are involved in the replication and infection process of coronaviruses. In conclusion, the study of cellular lipids and remodeling of lipid metabolism in coronavirus infections provides a good background for the development of antiviral drugs and vaccines.
Conclusion
-
Ethics Approval
Not applicable.
-
Conflicts of Interest
The authors have no conflicts of interest to declare.
-
Funding
None.
-
Availability of Data
All data generated or analyzed during this study are included in this published article. For other data, these may be requested through the corresponding author.
-
Authors’ contributions
Conceptualization: MZ; Data curation: PH, SS, AR; Investigation: PH, SS; Supervision: MZ; Writing–original draft: SN, MM, FB; Writing–review & editing: MZ, PH.
-
Additional Contributions
The images that constitute Figure 1 were provided by Milad Zandi (Tehran University of Medical Sciences, Tehran, Iran).
Article information
| Virus | Receptor | Steps of the coronavirus life cycle | Lipid interactions | Endocytic pathway | References |
|---|---|---|---|---|---|
| CCoV serotype 2 | APN | Entry | Plasma membrane cholesterol | Unknown | [54,55] |
| MHV | CEACAM1 | Entry | Lipid rafts | Clathrin-mediated endocytosis | [33,48,49] |
| FCoV serotype 2 | APN | Entry | Cholesterol | Clathrin- and caveolae-independent pathway that depends strongly on dynamin | [33,54] |
| PEDV | APN | Entry | Cholesterol, lipid rafts | Clathrin- and caveolae-mediated endocytosis pathways | [33,56] |
| TGEV | APN | Entry | Cholesterol | The route of entry is not yet known in terms of which endocytosis pathway occurs (clathrin- or non-clathrin-dependent mechanism or both) | [57,58] |
| IBV | Not recognized | Entry | Lipid rafts | Macropinocytosis | [45] |
| HCoV-229E | APN | 1. Entry | 1. Cholesterol | Caveolae-dependent endocytic | [44,49,53,59] |
| 2. Replication | 2. LA and AA | ||||
| HCoV-OC43 | HLA class I, sialic acids, Neu5,9 | Entry | Cholesterol | Caveolae- dependent endocytosis | [33,51] |
| MERS-CoV | DPP4 or CD26 | Replication | LA and AA | Clathrin-mediated endocytosis | [51,53,60] |
| SARS-CoV | ACE2 | Entry | Lipid rafts | Clathrin- and caveolae-independent mechanism; may involve a clathrin-mediated or clathrin-dependent mechanism | [46,50] |
CCoV, canine coronavirus; APN, aminopeptidase N; MHV, murine hepatitis virus; CEACAM1, carcinoembryonic antigen-related cell adhesion molecule 1; FCoV, feline coronavirus; PEDV, porcine epidemic diarrhea virus; TGEV, transmissible gastroenteritis virus; IBV, infectious bronchitis virus; HCoV-229E, human coronavirus 229E; LA, linoleic acid; AA, arachidonic acid; HCoV-OC43, human coronavirus OC43; HLA, human leukocyte antigen; MERS-CoV, Middle East respiratory syndrome coronavirus; DPP4, dipeptidyl peptidase 4; SARS-CoV, severe acute respiratory syndrome coronavirus; ACE2, angiotensin-converting enzyme-2.
- 1. Han Y, Du J, Su H, et al. Identification of diverse bat alphacoronaviruses and betacoronaviruses in China provides new insights into the evolution and origin of coronavirus-related diseases. Front Microbiol 2019;10:1900. ArticlePubMedPMC
- 2. Bermingham A, Chand MA, Brown CS, et al. Severe respiratory illness caused by a novel coronavirus, in a patient transferred to the United Kingdom from the Middle East, September 2012. Euro Surveill 2012;17:20290. ArticlePubMed
- 3. Hui DS, I Azhar E, Madani TA, et al. The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health: the latest 2019 novel coronavirus outbreak in Wuhan, China. Int J Infect Dis 2020;91:264−6.ArticlePubMedPMC
- 4. Bosch BJ, Martina BE, Van Der Zee R, et al. Severe acute respiratory syndrome coronavirus (SARS-CoV) infection inhibition using spike protein heptad repeat-derived peptides. Proc Natl Acad Sci U S A 2004;101:8455−60.ArticlePubMedPMC
- 5. Memish ZA, Mishra N, Olival KJ, et al. Middle East respiratory syndrome coronavirus in bats, Saudi Arabia. Emerg Infect Dis 2013;19:1819−23.ArticlePubMedPMC
- 6. Mobini Kesheh M, Shavandi S, Hosseini P, et al. Bioinformatic HLA studies in the context of SARS-CoV-2 pandemic and review on association of HLA alleles with preexisting medical conditions. Biomed Res Int 2021;2021:6693909. ArticlePubMedPMC
- 7. Millan-Onate J, Rodriguez-Morales AJ, Camacho-Moreno G, et al. A new emerging zoonotic virus of concern: the 2019 novel Coronavirus (SARS CoV-2). Infectio 2020;24:187−92.
- 8. Poortahmasebi V, Zandi M, Soltani S, et al. Clinical performance of RT-PCR and chest CT scan for COVID-19 diagnosis; a systematic review. Adv J Emerg Med 2020;4(2s):e57.
- 9. World Health Organization (WHO). Coronavirus disease (COVID-19) weekly epidemiological update and weekly operational update [Internet]. Geneva: WHO; 2021 [cited 2021 Sep 14]. Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports.
- 10. Villanueva RA, Rouille Y, Dubuisson J. Interactions between virus proteins and host cell membranes during the viral life cycle. Int Rev Cytol 2005;245:171−244.ArticlePubMedPMC
- 11. Wu F, Zhao S, Yu B, et al. A new coronavirus associated with human respiratory disease in China. Nature 2020;579:265−9.ArticlePubMedPMC
- 12. Heaton NS, Randall G. Multifaceted roles for lipids in viral infection. Trends Microbiol 2011;19:368−75.ArticlePubMedPMC
- 13. Thaker SK, Ch’ng J, Christofk HR. Viral hijacking of cellular metabolism. BMC Biol 2019;17:59. ArticlePubMedPMC
- 14. Martin-Acebes MA, Vazquez-Calvo A, Caridi F, et al. Lipid involvement in viral infections: present and future perspectives for the design of antiviral strategies. In: Baez RV, editor. Lipid metabolism. Rijeka: InTech; 2013. p. 291-322.
- 15. Sun X, Whittaker GR. Role for influenza virus envelope cholesterol in virus entry and infection. J Virol 2003;77:12543−51.ArticlePubMedPMC
- 16. Nagy PD, Strating JR, van Kuppeveld FJ. Building viral replication organelles: close encounters of the membrane types. PLoS Pathog 2016;12:e1005912.ArticlePubMedPMC
- 17. Hsu NY, Ilnytska O, Belov G, et al. Viral reorganization of the secretory pathway generates distinct organelles for RNA replication. Cell 2010;141:799−811.ArticlePubMedPMC
- 18. Diamond DL, Syder AJ, Jacobs JM, et al. Temporal proteome and lipidome profiles reveal hepatitis C virus-associated reprogramming of hepatocellular metabolism and bioenergetics. PLoS Pathog 2010;6:e1000719.ArticlePubMedPMC
- 19. Fecchi K, Anticoli S, Peruzzu D, et al. Coronavirus interplay with lipid rafts and autophagy unveils promising therapeutic targets. Front Microbiol 2020;11:1821. ArticlePubMedPMC
- 20. Kraft ML. Sphingolipid organization in the plasma membrane and the mechanisms that influence it. Front Cell Dev Biol 2017;4:154. ArticlePubMedPMC
- 21. Takahashi T, Suzuki T. Function of membrane rafts in viral lifecycles and host cellular response. Biochem Res Int 2011;2011:245090. ArticlePubMedPMC
- 22. Mazzon M, Mercer J. Lipid interactions during virus entry and infection. Cell Microbiol 2014;16:1493−502.ArticlePubMedPMC
- 23. Chazal N, Gerlier D. Virus entry, assembly, budding, and membrane rafts. Microbiol Mol Biol Rev 2003;67:226−37.ArticlePubMedPMC
- 24. Barman S, Nayak DP. Lipid raft disruption by cholesterol depletion enhances influenza A virus budding from MDCK cells. J Virol 2007;81:12169−78.ArticlePubMedPMC
- 25. Bajimaya S, Frankl T, Hayashi T, et al. Cholesterol is required for stability and infectivity of influenza A and respiratory syncytial viruses. Virology 2017;510:234−41.ArticlePubMed
- 26. Snyers L, Zwickl H, Blaas D. Human rhinovirus type 2 is internalized by clathrin-mediated endocytosis. J Virol 2003;77:5360−9.ArticlePubMedPMC
- 27. Grassme H, Riehle A, Wilker B, et al. Rhinoviruses infect human epithelial cells via ceramide-enriched membrane platforms. J Biol Chem 2005;280:26256−62.ArticlePubMed
- 28. Grassi G, Di Caprio G, Fimia GM, et al. Hepatitis C virus relies on lipoproteins for its life cycle. World J Gastroenterol 2016;22:1953−65.ArticlePubMedPMC
- 29. Cascella M, Rajnik M, Aleem A, et al. Features, evaluation, and treatment of coronavirus (COVID-19) [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 [cited 2021 Sep 20]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK554776/.
- 30. Fehr AR, Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol 2015;1282:1−23.ArticlePubMedPMC
- 31. Dhama K, Khan S, Tiwari R, et al. Coronavirus Disease 2019-COVID-19. Clin Microbiol Rev 2020;33:e00028−20.ArticlePubMedPMC
- 32. Tang X, Wu C, Li X, et al. On the origin and continuing evolution of SARS-CoV-2. Natl Sci Rev 2020;7:1012−23.ArticlePubMedPMC
- 33. Belouzard S, Millet JK, Licitra BN, et al. Mechanisms of coronavirus cell entry mediated by the viral spike protein. Viruses 2012;4:1011−33.ArticlePubMedPMC
- 34. Dawood AA, Altobje MA. Inhibition of N-linked glycosylation by tunicamycin may contribute to the treatment of SARS-CoV-2. Microb Pathog 2020;149:104586. ArticlePubMedPMC
- 35. Spiga O, Bernini A, Ciutti A, et al. Molecular modelling of S1 and S2 subunits of SARS coronavirus spike glycoprotein. Biochem Biophys Res Commun 2003;310:78−83.ArticlePubMedPMC
- 36. de Haan CA, de Wit M, Kuo L, et al. The glycosylation status of the murine hepatitis coronavirus M protein affects the interferogenic capacity of the virus in vitro and its ability to replicate in the liver but not the brain. Virology 2003;312:395−406.ArticlePubMed
- 37. Fung TS, Liu DX. Post-translational modifications of coronavirus proteins: roles and function. Future Virol 2018;13:405−30.ArticlePubMedPMC
- 38. Kuo L, Hurst-Hess KR, Koetzner CA, et al. Analyses of coronavirus assembly interactions with interspecies membrane and nucleocapsid protein chimeras. J Virol 2016;90:4357−68.ArticlePubMedPMC
- 39. Fung TS, Liao Y, Liu DX. Regulation of stress responses and translational control by coronavirus. Viruses 2016;8:184. ArticlePubMedPMC
- 40. McBride R, van Zyl M, Fielding BC. The coronavirus nucleocapsid is a multifunctional protein. Viruses 2014;6:2991−3018.ArticlePubMedPMC
- 41. Simons K, Sampaio JL. Membrane organization and lipid rafts. Cold Spring Harb Perspect Biol 2011;3:a004697. ArticlePubMedPMC
- 42. Kesheh MM, Hosseini P, Soltani S, et al. An overview on the seven pathogenic human coronaviruses. Rev Med Virol 2021 Aug 2 [Epub]. https://doi.org/10.1002/rmv.2282.Article
- 43. Baglivo M, Baronio M, Natalini G, et al. Natural small molecules as inhibitors of coronavirus lipid-dependent attachment to host cells: a possible strategy for reducing SARS-COV-2 infectivity? Acta Biomed 2020;91:161−4.PubMedPMC
- 44. Nomura R, Kiyota A, Suzaki E, et al. Human coronavirus 229E binds to CD13 in rafts and enters the cell through caveolae. J Virol 2004;78:8701−8.ArticlePubMedPMC
- 45. Guo H, Huang M, Yuan Q, et al. The important role of lipid raft-mediated attachment in the infection of cultured cells by coronavirus infectious bronchitis virus Beaudette strain. PLoS One 2017;12:e0170123.ArticlePubMedPMC
- 46. Lu Y, Liu DX, Tam JP. Lipid rafts are involved in SARS-CoV entry into Vero E6 cells. Biochem Biophys Res Commun 2008;369:344−9.ArticlePubMedPMC
- 47. Glende J, Schwegmann-Wessels C, Al-Falah M, et al. Importance of cholesterol-rich membrane microdomains in the interaction of the S protein of SARS-coronavirus with the cellular receptor angiotensin-converting enzyme 2. Virology 2008;381:215−21.ArticlePubMed
- 48. Choi KS, Aizaki H, Lai MM. Murine coronavirus requires lipid rafts for virus entry and cell-cell fusion but not for virus release. J Virol 2005;79:9862−71.ArticlePubMedPMC
- 49. Burkard C, Verheije MH, Wicht O, et al. Coronavirus cell entry occurs through the endo-/lysosomal pathway in a proteolysis-dependent manner. PLoS Pathog 2014;10:e1004502.ArticlePubMedPMC
- 50. Wang H, Yang P, Liu K, et al. SARS coronavirus entry into host cells through a novel clathrin- and caveolae-independent endocytic pathway. Cell Res 2008;18:290−301.ArticlePubMed
- 51. Owczarek K, Szczepanski A, Milewska A, et al. Early events during human coronavirus OC43 entry to the cell. Sci Rep 2018;8:7124. ArticlePubMedPMC
- 52. Van Hamme E, Dewerchin HL, Cornelissen E, et al. Clathrin- and caveolae-independent entry of feline infectious peritonitis virus in monocytes depends on dynamin. J Gen Virol 2008;89(Pt 9). 2147−56.ArticlePubMed
- 53. Yan B, Chu H, Yang D, et al. Characterization of the lipidomic profile of human coronavirus-infected cells: implications for lipid metabolism remodeling upon coronavirus replication. Viruses 2019;11:73. ArticlePubMedPMC
- 54. Le Poder S. Feline and canine coronaviruses: common genetic and pathobiological features. Adv Virol 2011;2011:609465. ArticlePubMedPMC
- 55. Pratelli A, Colao V. Role of the lipid rafts in the life cycle of canine coronavirus. J Gen Virol 2015;96(Pt 2):331−7.Article
- 56. Wei X, She G, Wu T, et al. PEDV enters cells through clathrin-, caveolae-, and lipid raft-mediated endocytosis and traffics via the endo-/lysosome pathway. Vet Res 2020;51:10. ArticlePubMedPMC
- 57. Hansen GH, Delmas B, Besnardeau L, et al. The coronavirus transmissible gastroenteritis virus causes infection after receptor-mediated endocytosis and acid-dependent fusion with an intracellular compartment. J Virol 1998;72:527−34.ArticlePubMedPMC
- 58. Yin J, Glende J, Schwegmann-Wessels C, et al. Cholesterol is important for a post-adsorption step in the entry process of transmissible gastroenteritis virus. Antiviral Res 2010;88:311−6.ArticlePubMedPMC
- 59. Kawase M, Shirato K, Matsuyama S, et al. Protease-mediated entry via the endosome of human coronavirus 229E. J Virol 2009;83:712−21.ArticlePubMed
- 60. Yang N, Shen HM. Targeting the endocytic pathway and autophagy process as a novel therapeutic strategy in COVID-19. Int J Biol Sci 2020;16:1724−31.ArticlePubMedPMC
- 61. Snijder EJ, van der Meer Y, Zevenhoven-Dobbe J, et al. Ultrastructure and origin of membrane vesicles associated with the severe acute respiratory syndrome coronavirus replication complex. J Virol 2006;80:5927−40.ArticlePubMedPMC
- 62. de Castro IF, Volonte L, Risco C. Virus factories: biogenesis and structural design. Cell Microbiol 2013;15:24−34.ArticlePubMed
- 63. den Boon JA, Ahlquist P. Organelle-like membrane compartmentalization of positive-strand RNA virus replication factories. Annu Rev Microbiol 2010;64:241−56.ArticlePubMed
- 64. Romero-Brey I, Bartenschlager R. Membranous replication factories induced by plus-strand RNA viruses. Viruses 2014;6:2826−57.ArticlePubMedPMC
- 65. Nicora CD, Sims AC, Bloodsworth KJ, et al. Metabolite, protein, and lipid extraction (MPLEx): a method that simultaneously inactivates Middle East respiratory syndrome coronavirus and allows analysis of multiple host cell components following infection. In: Vijay R, editors. MERS coronavirus: methods and protocols. New York: Humana; 2020. p. 173-94.
- 66. Oudshoorn D, Rijs K, Limpens RW, et al. Expression and cleavage of Middle East respiratory syndrome coronavirus nsp3-4 polyprotein induce the formation of double-membrane vesicles that mimic those associated with coronaviral RNA replication. mBio 2017;8:e01658−17.ArticlePubMedPMC
- 67. den Boon JA, Diaz A, Ahlquist P. Cytoplasmic viral replication complexes. Cell Host Microbe 2010;8:77−85.ArticlePubMedPMC
- 68. Angelini MM, Akhlaghpour M, Neuman BW, et al. Severe acute respiratory syndrome coronavirus nonstructural proteins 3, 4, and 6 induce double-membrane vesicles. mBio 2013;4:e00524−13.Article
- 69. Muller C, Hardt M, Schwudke D, et al. Inhibition of cytosolic phospholipase A2α impairs an early step of coronavirus replication in cell culture. J Virol 2018;92:e01463−17.ArticlePubMedPMCPDF
- 70. Fernandez-Oliva A, Ortega-Gonzalez P, Risco C. Targeting host lipid flows: exploring new antiviral and antibiotic strategies. Cell Microbiol 2019;21:e12996.ArticlePubMedPMC
- 71. Verma SP. HIV: a raft-targeting approach for prevention and therapy using plant-derived compounds (review). Curr Drug Targets 2009;10:51−9.ArticlePubMed
- 72. Leneva I, Kartashova N, Poromov A, et al. Antiviral activity of umifenovir in vitro against a broad spectrum of coronaviruses, including the novel SARS-CoV-2 virus. Viruses 2021;13:1665. ArticlePubMedPMC
- 73. Orlowski S, Mourad JJ, Gallo A, et al. Coronaviruses, cholesterol and statins: involvement and application for COVID-19. Biochimie 2021;189:51−64.ArticlePubMedPMC
References
Figure & Data
References
Citations

- PEDV inhibits HNRNPA3 expression by miR-218-5p to enhance cellular lipid accumulation and promote viral replication
Xiaojie Shi, Qi Zhang, Naling Yang, Quanqiong Wang, Yanxia Zhang, Xingang Xu, Xiang-Jin Meng, Ying Fang
mBio.2024;[Epub] CrossRef - Orsay Virus Infection of Caenorhabditis elegans Is Modulated by Zinc and Dependent on Lipids
Luis Alberto Casorla-Perez, Ranya Guennoun, Ciro Cubillas, Bo Peng, Kerry Kornfeld, David Wang, Rebecca Ellis Dutch
Journal of Virology.2022;[Epub] CrossRef


 PubReader
PubReader ePub Link
ePub Link Cite
Cite